Gut-Brain-Libido Axis: The Role Of Serotonin, Probiotics And Key Nutrients In Intimacy
A Deep‑Dive into the Gut‑Brain‑Intimacy Connection
People often ask, “Can nutrition or even probiotics boost my libido?” The answer isn’t a simple “yes” or “no.” It lives within a web of biochemical pathways that connect the gut, brain, immune system, and even our sleep cycles. Understanding these connections lets you craft nutrition‑based strategies that support desire, pleasure, and emotional closeness — the very foundations of a healthy intimate life.
Talking about libido and intimacy, whether with a partner or a practitioner, is part of taking your sexual health as seriously as any other aspect of well-being, and:
It improves relationships and satisfaction. Open, honest communication about needs, boundaries and desires is strongly linked with higher sexual satisfaction, emotional closeness and a stronger bond between partners.
It reduces shame and isolation. Naming sexual worries out loud helps challenge the idea that they are “taboo” or uniquely yours, and creates space for evidence‑based, compassionate support instead of silent avoidance.
It protects overall health. Sexual health is tied to mental health, cardiovascular health, immune function and quality of life, so voicing changes in libido or intimacy can act as an early-warning sign that something in the wider system needs attention.
Serotonin: More Than a “Feel‑Good” Molecule
How Gut‑Derived Serotonin Talks to the Brain
Vagal Afferents:
EC cells release 5‑HT that activates receptors on vagal afferents, sending signals to the nucleus tractus solitarius (brainstem structure that acts as the primary sensory relay station for autonomic, visceral, and taste information) and then to limbic structures governing desire.
N.B. Vagal afferents are sensory neurones of the vagus nerve that transmit information from visceral organs (gut, heart, lungs) to the brain, forming a crucial pathway in the gut-brain axis. They monitor mechanical, chemical, and immune signals to regulate satiety, digestion, and homeostasis. Dysfunction in these pathways is linked to obesity, anxiety, and inflammation.
Enteroendocrine Crosstalk:
5‑HT stimulates the secretion of GLP‑1 and PYY, hormones that can cross the blood‑brain barrier (BBB) and modulate mood.
Circulating Metabolites:
Some gut microbes convert tryptophan into indole‑propionic acid, a neuroprotective compound that indirectly supports serotonergic tone (i.e., activity and availability of serotonin).
A 2023 human‑microbiome trial showed that probiotic supplementation with Bifidobacterium longum increased plasma 5‑HT metabolites and improved self‑reported sexual satisfaction scores.
This is what researchers said about it:
“Pathophysiological mechanisms involved in sexual dysfunction are closely linked to the gut microbiota, a crucial regulator of metabolism, immunity, and overall health. Dysbiosis, or imbalance in the gut microbiota, is associated with metabolic disorders, including type 2 diabetes. The gut microbiota produces metabolites such as short-chain fatty acids (SCFAs) that interact with the nervous, immune, and metabolic systems, impacting systemic health.
Recent research has identified the gut–brain axis as a key pathway through which gut microbiota influences sexual function by regulating neural signalling and hormone metabolism. Specifically, the gut microbiota plays a critical role in modulating sex hormones such as oestrogen and testosterone, which are essential for maintaining sexual health. In diabetic individuals, dysbiosis exacerbates sexual dysfunction through mechanisms including increased inflammation, oxidative stress, and impaired vascular function, all of which are influenced by the gut microbiota.”
“Bifidobacterium spp. and Lactobacillus spp. produce GABA, an inhibitory neurotransmitter involved in stress regulation, whereas Escherichia spp., Streptococcus spp., Candida spp., and Bacillus spp. contribute to serotonin and dopamine biosynthesis. These microbial-derived neurotransmitters play a fundamental role in pleasure, motivation, and sexual reward pathways. Although their direct impact on the brain is limited due to the selective permeability of the BBB, they exert indirect effects via interactions with the enteric nervous system and vagus nerve signalling.
Moreover, certain bacterial strains, such as Bifidobacterium infantis, enhance circulating tryptophan levels, leading to increased serotonin availability in the brain, which is associated with improvements in mood and sexual motivation.”
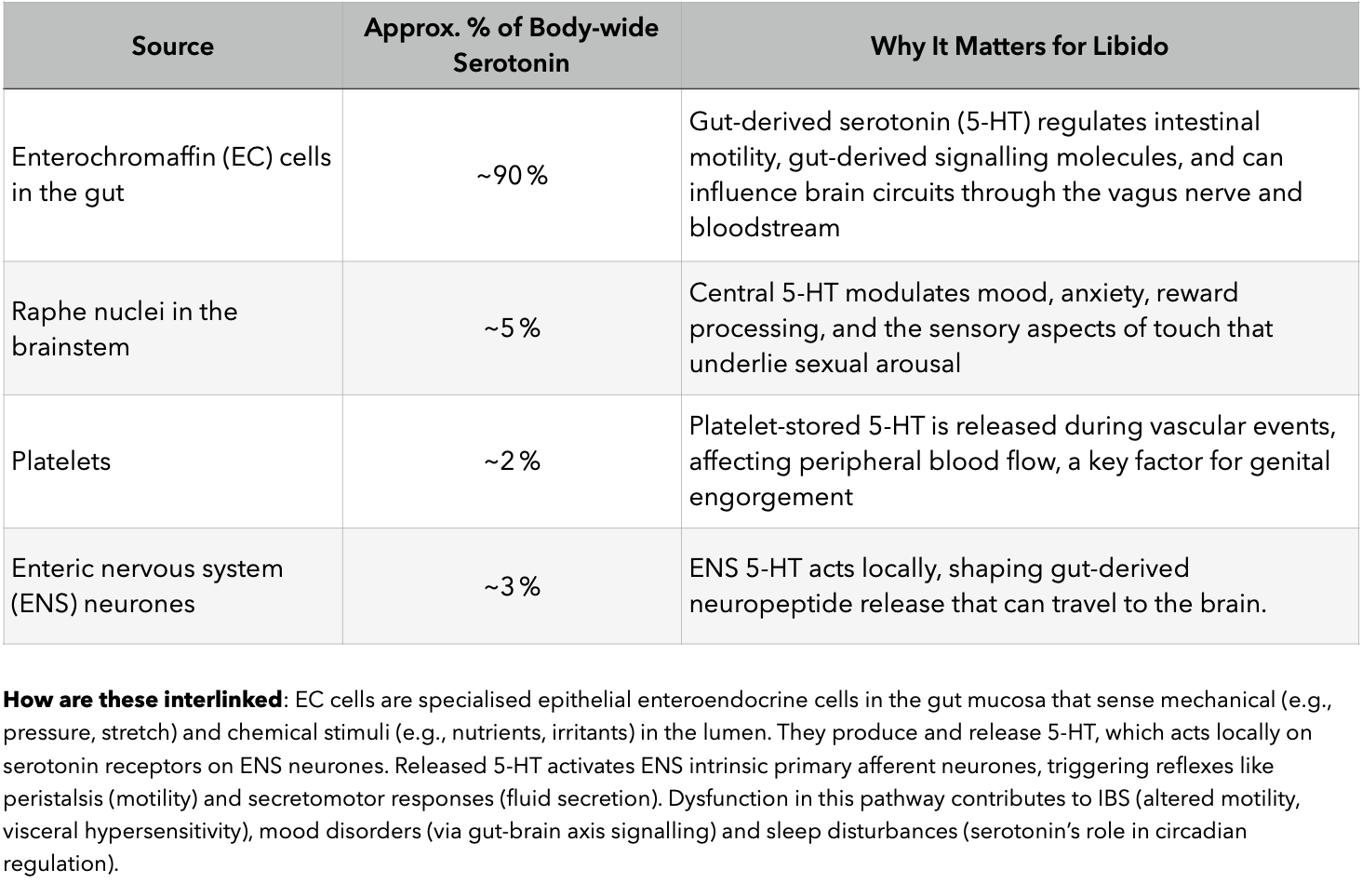
In a nutshell
Gut-derived serotonin (5-HT), produced primarily by enterochromaffin (EC) cells, communicates with the brain via multiple pathways, influencing mood, appetite, stress and even aspects of desire through the gut-brain axis.
Vagal Afferents Pathway
EC cells release 5-HT in response to luminal stimuli, activating 5-HT receptors on vagal afferent nerve terminals in the gut wall.
These signals travel to the brain, reaching limbic structures like the amygdala, hypothalamus and prefrontal cortex, modulating emotional processing, motivation and reward pathways potentially linked to desire and intimacy.
Enteroendocrine Crosstalk
Gut 5-HT from EC cells can stimulate nearby enteroendocrine cells (EECs), promoting release of peptides like glucagon-like peptide-1 (GLP-1) and peptide YY (PYY).
GLP-1 and PYY influence central serotonin systems and cross or signal across the blood-brain barrier (BBB) to regulate mood, satiety and hedonic responses.
5-HT modulates inflammation and EEC secretion, indirectly supporting mood via reduced neuroinflammation.
Circulating Metabolites
Gut microbes metabolise dietary tryptophan (precursor to 5-HT) into indole derivatives like indole-3-propionic acid (IPA, or indole-propionic acid). IPA is neuroprotective, crosses the BBB, and modulates serotonergic pathways and reduces oxidative stress, thereby supporting central 5-HT tone and mood stability.
— These pathways interconnect: vagal signalling provides rapid neural relay, while hormonal/metabolite routes offer slower, sustained effects, collectively linking gut 5-HT dysregulation to mood/libido issues.
Probiotics: Do They Really Impact Libido?
Probiotics are emerging as a promising adjunct for sexual dysfunction, particularly in cases linked to medications like selective serotonin reuptake inhibitors (SSRIs), which often disrupt libido via serotonin imbalances.
A 2024 double-blind randomised trial found that women with depression on SSRIs who added a probiotic blend (including Lactobacillus acidophilus and Bifidobacterium bifidum) for 8 weeks showed significant improvements in Female Sexual Function Index (FSFI) scores, across desire, arousal, lubrication, orgasm, satisfaction and pain, compared to SSRIs alone. Depressive symptoms also eased notably.
— Mechanisms at Play
Probiotics restore gut microbiome balance, reducing systemic inflammation and improving gut-derived serotonin production, which signals via the gut-brain axis to support mood and sexual motivation. They modulate sex hormones, including oestrogen and testosterone, and their antioxidant effects help combat oxidative stress in reproductive tissues.
While several medical organisations increasingly note gut health’s role in sexual function (e.g., via microbiome-hormone links), formal guidelines lag behind, calling for more trials to integrate probiotics routinely.
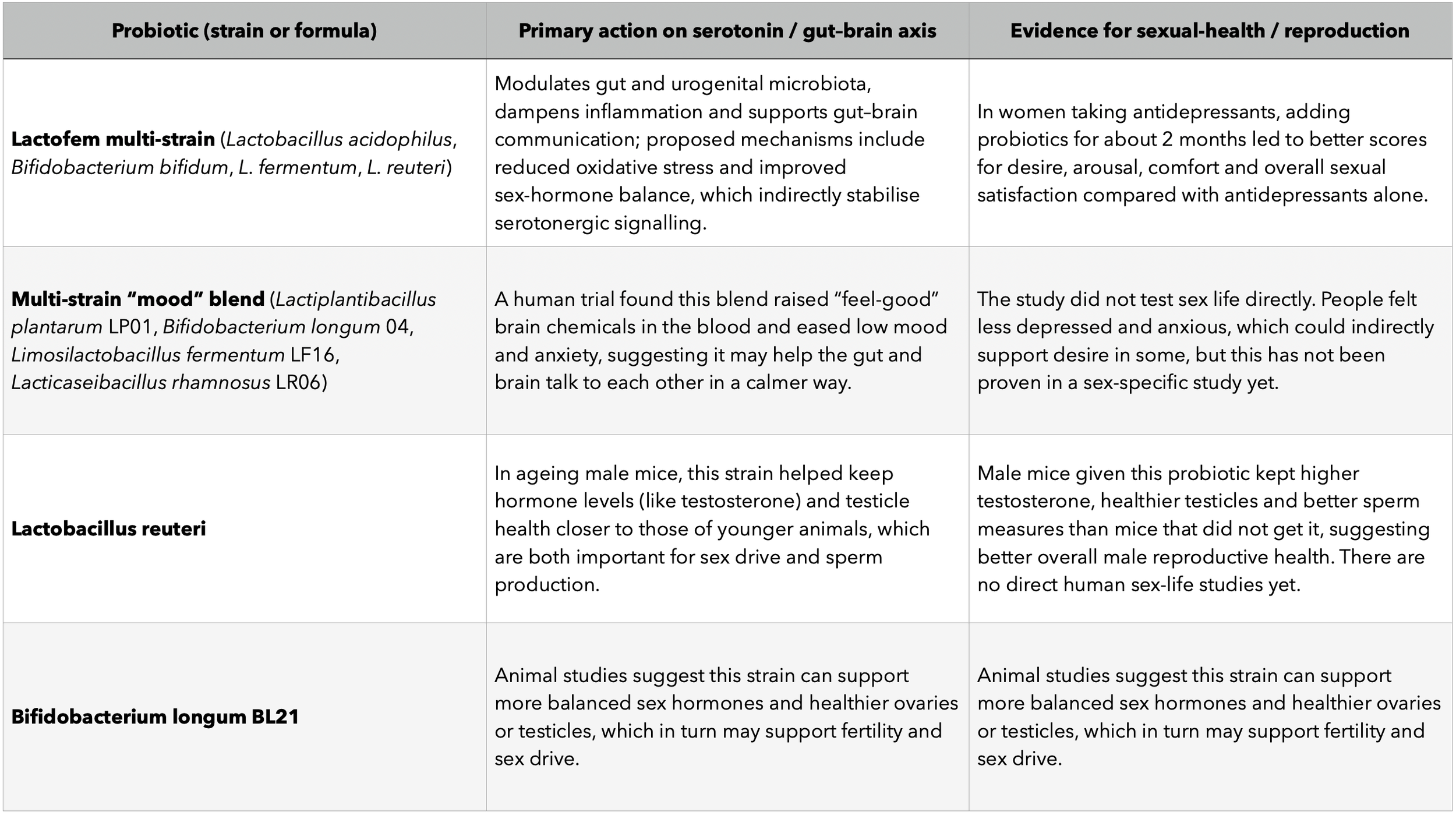
Several other probiotic products have been tested in humans for sexual function or fertility:
Lactofem in women with PCOS on letrozole. Women with polycystic ovary syndrome who took Lactofem together with letrozole for 8 weeks had better sexual‑function scores and higher pregnancy rates than women on letrozole alone.
Lactofem in women on antidepressants (SSRIs) . Depressed women taking SSRIs who added Lactofem for 8 weeks reported greater improvements in sexual satisfaction and overall sexual‑function scores than women taking SSRIs alone.
A formula for men with fertility problems. A multi‑strain probiotic called FamiLact improved sperm count, movement and shape over about 80 days compared with a placebo, which is relevant for male fertility, even though desire/erections were not directly measured.
Single‑strain Lactobacillus acidophilus in midlife women. In pre‑ and post‑menopausal women with sexual difficulties, 8 weeks of Lactobacillus acidophilus capsules led to higher total sexual‑function scores and better quality of life than placebo.
General probiotic mix in post‑menopausal women .A recent trial in post‑menopausal women found that 6 weeks of a daily probiotic capsule improved fatigue and sexual‑function scores compared with placebo.
A 2025 systematic review pulled these and similar trials together and concluded that probiotics can meaningfully improve sexual‑function scores in women in several settings (antidepressant use, PCOS, menopause), and can improve sperm quality in men, although direct libido/erection outcomes in men are still scarce.
Nutrition That Fuels Serotonin‑Intimacy
Everyday nutrients that support serotonin:
Tryptophan (amino acid)
Your body uses tryptophan to make serotonin, the “feel‑good” brain chemical that supports mood, sleep and calm.
Food sources: turkey, chicken, eggs, cheese, chickpeas, pumpkin seeds, nuts, oats.
Practical tip: combine a tryptophan‑rich food with a healthy carb (like oats or whole‑grain toast) so more tryptophan can reach your brain.
Omega‑3 fats (EPA/DHA)
Help keep brain cells flexible so serotonin signals travel smoothly, and also calm inflammation that can drag mood down.
Food sources: Salmon, mackerel, sardines, trout, algae oil, walnuts, chia and flaxseeds.
Practical tip: Eat wild oily fish 2–3 times per week or talk to a practitioner about a fish or algae oil supplement that gives around 1 g EPA + DHA per day.
Vitamin B6
Needed in the step where your body turns tryptophan into serotonin; low B6 can make that process less efficient.
Food sources: chickpeas, potatoes, bananas, poultry, fish, nuts, seeds.
Practical tip: include at least one B6‑rich food daily (for example, a banana, some hummus, or a portion of chicken or fish).
Magnesium
Supports a calmer nervous system and works alongside serotonin and other relaxing brain chemicals.
Food sources: dark leafy greens, pumpkin seeds, nuts, whole grains, beans, dark chocolate.
Practical tip: add magnesium‑rich foods most days (e.g. big green salads with seeds, nuts as snacks, a little dark chocolate) and seek advice before using high‑dose supplements.
Plant pigments (polyphenols)
Help reduce irritation and stress in the body and support a healthier gut environment, which can, in turn, support mood.
Food sources: berries, colourful fruits and vegetables, green tea, herbs, and spices.
Practical tip: “Eat the rainbow.” Add at least one brightly coloured fruit or veg or a cup of green/herbal tea every day. In addition to one portion of the deepest green organic vegetable (brassicas).
Prebiotic fibre
Feeds friendly gut bacteria, which make helpful substances that support gut and brain health.
Food sources: onions, garlic, leeks, chicory root, Jerusalem artichokes, oats, beans, lentils, cooked‑and‑cooled potatoes or rice.
Practical tip: build up fibre slowly towards 15–25 g per day from whole plant foods, and drink enough water so your digestion stays comfortable.
Overall: eating these nutrients regularly gives your body the raw materials and support it needs to make and use serotonin, which can help with mood, stress resilience and, for many people, enjoyment of touch and intimacy.
Beyond Serotonin: Other Tissues & Mediators That Shape Desire
Dopamine‑Rich Areas (Ventral Tegmental Area & Nucleus Accumbens)
Dopamine drives reward and incentive salience; gut microbes that increase short‑chain fatty acids (SCFAs) can upregulate dopaminergic signalling.
Endocannabinoid System
Gut‑derived N‑acylethanolamines* interact with CB₁ receptors in the brain, influencing pleasure and stress resilience.
Peripheral Nitric Oxide (NO)
Produced by endothelial cells; essential for genital vasodilation. Gut inflammation can impair NO synthase activity, dampening erectile response, and so can hyperhomocysteinemia. Homocysteine is a sulfur-containing amino acid produced during protein metabolism, typically converted into other essential amino acids using Vitamin B12, B6, and folate. Elevated levels are a risk factor for cardiovascular disease, blood clots, stroke, and cognitive decline.
—
* In simple terms: Your body makes its own cannabis‑like substances, called endocannabinoids, which act on CB1 receptors in the brain. These receptors are involved in pleasure, reward, and coping with stress. Some of these cannabis‑like molecules are N‑acylethanolamines (fatty compounds that can be produced in the gut and elsewhere in the body). When these molecules reach the brain and activate CB1 receptors, they can tune how much pleasure you feel and how resilient you are to stress, a bit like a dimmer switch for reward, motivation and calm.
How Stress, GABA‑Glutamate Balance, and Inflammation Derail Libido
Mechanisms:
Ongoing high stress (chronically raised cortisol)
Long‑term stress hormones can interfere with brain signals that make serotonin, the “feel‑good” chemical, which can lower mood and desire.
Cortisol also blunts the normal pulse of hormones from the brain that drive ovulation, testosterone and overall sex‑hormone balance, which can dampen libido.
Imbalance between calming (GABA) and stimulating (glutamate) brain signals
Too much glutamate (the “accelerator”) and not enough GABA (the “brake”) can leave the brain in a wired, anxious state, which is not favourable for arousal or relaxation with a partner.
Over‑activation of these stress‑linked circuits in areas that control hormones and behaviour can reduce natural sexual interest and response.
Systemic inflammation
Inflammatory messenger molecules (like IL‑6 and TNF‑α) push the amino acid tryptophan away from making serotonin and into a different pathway (kynurenine), which is often linked with low mood and fatigue.
With less tryptophan available for serotonin, mood, motivation and pleasure can all drop, indirectly lowering libido.
Poor gut health and sluggish detox/elimination
When the gut lining is irritated, and bacterial balance is off, bacterial fragments (like LPS, an “endotoxin”) can leak into the bloodstream and trigger inflammation in the brain and body.
This “neuro‑inflammation” can disturb brain signalling, reduce nitric oxide (needed for healthy blood flow and erections) and make you feel more tired, flat and disconnected sexually.
Sleep, the Glymphatic System, and Sexual Vitality
Glymphatic clearance peaks during deep non‑REM sleep, flushing metabolic waste (including excess glutamate and inflammatory mediators) from the brain.
Chronic sleep restriction diminishes glymphatic flow, leading to accumulated neurotoxic proteins that blunt serotonergic and dopaminergic transmission.
Study highlight: Participants sleeping less than 6 hours per night for 2 weeks showed a 22 % drop in plasma 5‑HT levels and reported lower libido scores.
Putting It All Together – A 6‑Step Action Plan
1. Eat enough tryptophan each day
Include a decent portion of tryptophan‑rich foods daily, such as turkey or chicken, eggs, chickpeas, tofu, pumpkin seeds or nuts.
Combine them with a healthy carbohydrate (like brown rice, quinoa, or whole‑grain bread) to help more tryptophan reach your brain and support serotonin, mood and desire.
2. Consider a targeted probiotic
Use a quality multi‑strain probiotic that includes well‑researched strains from the Lactobacillus and Bifidobacterium families (for example, blends used in women on antidepressants or in PCOS trials), rather than relying on a single generic strain.
These can help calm gut inflammation, support hormone balance and improve mood and sexual‑function scores in some women, especially when low mood or medication side effects are part of the picture.
3. Feed your gut bacteria
Aim for plenty of fibre from plants each day (vegetables, pulses, oats, nuts, seeds) plus some “prebiotic” foods like onions, garlic, leeks, chicory or resistant starches, including cooked‑and‑cooled potatoes, tiger nuts, green banana (plantain), etc.
Add colourful fruits and berries regularly for their plant compounds; together, these choices help your gut bugs make substances that support brain chemistry, energy and motivation.
4. Support your brain chemistry with key nutrients
Make sure you are getting enough magnesium (around 300–400 mg per day from food and, if needed, supplements), vitamin B6 (from chickpeas, bananas, poultry, fish) and omega‑3 fats (ideally around 1 g EPA + DHA per day from oily fish or a supplement, if appropriate for you).
These nutrients help your calming and stimulating brain chemicals stay in balance and support healthy serotonin signalling, which underpins mood, relaxation and sexual interest.
5. Actively manage daily stress
Build in simple stress‑relief habits, such as 5–10 minutes of slow, deep belly breathing, a short walk outside, or 20 minutes of moderate exercise most days.
If suitable for you, stress‑supportive herbs like ashwagandha and holy basil (Tulsi) can help lower perceived stress and cortisol, which in turn takes pressure off hormones and libido (best done with practitioner guidance).
6. Protect your sleep window
Aim for 7–9 hours in bed at night in a dark, cool room, with screens off for at least 30 minutes before sleep.
Good‑quality sleep helps your brain clear waste products, cool down inflammation, and reset serotonin and other hormones that influence mood, energy and sexual desire.
Frequently Asked Questions
Q1. Can a single probiotic capsule replace a balanced diet?
No. Probiotics are adjuncts that work best when the diet already provides ample dietary fibre, tryptophan, and micronutrients.
Q2. Are there any safety concerns with high‑dose probiotics?
Generally safe for healthy adults. Immunocompromised individuals should consult a clinician before starting high‑CFU formulations. It is always best to consult a professional before supplementing, as many products do not deliver on their label claims or contain “dead” ingredients.
Q3. How long does it take to notice a change in libido after starting these interventions?
Most clinical trials report 4‑8 weeks for measurable improvements in sexual satisfaction scores.
Q4. Does the gut produce the serotonin that directly reaches the brain?
Gut‑produced serotonin does not cross the BBB in large amounts, but it influences the brain indirectly via vagal signalling, hormone release, and modulation of peripheral tryptophan availability.
Bottom Line
Serotonin is a team sport. The gut supplies the raw material; the brain’s raphe nuclei and peripheral platelets deliver the final “feel‑good” signal that shapes touch perception and desire.
Probiotics can tip the balance toward a healthier gut‑brain dialogue, especially strains that increase tryptophan‑derived indoles and calm inflammation.
Nutrition, stress control, and restorative sleep are the three pillars that keep the serotonin‑dopamine‑endocannabinoid network firing on all cylinders.
By integrating targeted probiotics, serotonin‑friendly foods, and lifestyle habits that protect the glymphatic system, you create a robust physiological foundation for greater intimacy, pleasure, and overall sexual well‑being.
References
Houston, DS., Vanhoutte, PM. (1986). Serotonin and the vascular system. Role in health and disease, and implications for therapy. Drugs. 31(2), pp. 149-163. doi:10.2165/00003495-198631020-00004
Merino del Portillo, M., Clemente-Suárez, V. J., Ruisoto, P. et al. (2024). Nutritional Modulation of the Gut–Brain Axis: A Comprehensive Review of Dietary Interventions in Depression and Anxiety Management. Metabolites. 14(10), 549. doi:10.3390/metabo14100549
Marano, G., Anesini, MB., Milintenda, M. et al. (2025). Discovering a new paradigm: Gut microbiota as a central modulator of sexual health. World Journal of Gastrointestinal Pathophysiology. 16(3), 107823. doi:10.4291/wjgp.v16.i3.107823
Mörkl, S., Narrath, M., Schlotmann, D. et al. (2025). Multi-species probiotic supplement enhances vagal nerve function - results of a randomized controlled trial in patients with depression and healthy controls. Gut Microbes. 2025 Dec;17(1):2492377. doi:10.1080/19490976.2025.2492377
Oxenkrug, GF. (2010). Tryptophan kynurenine metabolism as a common mediator of genetic and environmental impacts in major depressive disorder: The serotonin hypothesis revisited 40 years later. Israel Journal of Psychiatry and Related Sciences. 47(1), pp. 56-63.
Palacios-Arellano, MP., Landa-García, JN., García-Martínez, ED. et al. (2025). Brain nuclei in the regulation of sexual behavior, peripheral nerves related to reproduction, and their alterations in neurodegenerative diseases: A brief review. Brain Science. 15(9), 942. doi:10.3390/brainsci15090942
Patrick, RP., Ames, BN. (2015). Vitamin D and the omega-3 fatty acids control serotonin synthesis and action, part 2: Relevance for ADHD, bipolar disorder, schizophrenia, and impulsive behavior. FASEB Journal. 29(6), pp. 2207-2222. doi:10.1096/fj.14-268342
Pickering, G., Mazur, A., Trousselard, M. et al. (2020). Magnesium status and stress: The vicious circle concept revisited. Nutrients. 12(12), 3672. doi:10.3390/nu12123672
Yang, SJ., Nguyen, TTM., Jin, X. et al. (2025). A PRISMA systematic review of sexual dysfunction and probiotics with pathophysiological mechanisms. Biology (Basel). 14(3), 286. doi:10.3390/biology14030286