Detoxification: Key Roles of Magnesium. Should I Supplement with Magnesium?
Before, going deep into understanding the role of Magnesium in detoxification processes, let’s look at the role magnesium plays in our body, its importance to our health and its vital implementation in our diet.
Amongst other minerals including calcium, sodium, phosphorus, potassium and sulphur, magnesium form part of the macrominerals. While minerals such as: boron, chromium, copper, iron, manganese, selenium and zinc are called trace minerals.
What is the difference between a Macro Mineral and a Trace Mineral?
To answer this simple question, it is important to understand how food is classified once it has been ingested. Minerals, together with Vitamins, form a group called micronutrients. Protein, fat and carbohydrates form a group called macronutrients.
The difference between a macronutrient and a micronutrient is simple: while both groups are essential to the optimal functioning of our body, macronutrients are indeed the nutrients we desperately need to survive – and for energy, calculated in calories --, and we need lots of it (A diet -- based on 2000 calories -- should include 50-55% Carbohydrate (261 grams), 20-30% Fat (65 grams) and 18-30% Proteins (91 grams) depending on individual needs and physical activity). Micronutrients have no calorific value and do not contribute directly to energy needs; therefore, vitamins and minerals cannot be quantified in such percentage, and their value is calculated in micrograms (µg).
A macromineral should have a preponderant place in the diet (usually above 100 µg) and a trace mineral in infinitesimal quantity; although, It would be wrong to think that one is more important than the other.
What are Minerals?
Minerals are everywhere on the planet, and with the result of erosion, and other elements, minuscule particles of it are thrown everywhere on earth. Minerals become soluble and enter the soil. Plants, including vegetables and fruit trees, absorb those minerals, and through a chain reaction, are eaten by herbivores, that subsequently are eaten by carnivores. As humans, we place ourselves anywhere on this scale, ingesting minerals with all the food we eat.
It seems that it is unlikely to not meet the Recommended Daily Allowance (RDA), since all food contains some levels of Minerals.
Looking at the NHS and other countries’ social security deficit, it seems that we do not. This is explained by our modern lifestyle, our love for fast-and-easy food, snacking, and most of all — the result of food industrialisation — processed and refined food: highly calorific food, containing a large array of dangerous chemicals, lacking the vital nutrients our body needs, including minerals (refined sugar has 95% less magnesium) and fibre.
Despite the addition of micronutrients to the food, these are often present in a synthetic form and may not be absorbed by the body in the same way or not at all, the body unable to recognise the 'alien' man-made molecule.
What do Minerals do?
Minerals act as catalysts for many biological reactions within the body, including, muscle response, the transmission of messages through the nervous system, and the utilisation of nutrients in food.
Magnesium, too, has a specific action on our body. It is an important electrolyte needed for proper muscle, nerve, and enzyme function. It keeps heart rhythm steady, supports a healthy immune system and keeps bones strong. It also helps the body make and use energy and is needed to move other electrolytes (potassium and sodium) into and out of cells. Keeping in mind the osmosis process, the ideal ratio of magnesium, potassium and sodium must be kept at all times; a fall in the level of one will create an imbalance: often the start of many diseases.
Quick note:
A healthy cell allows nutrients to enter and be utilised, eliminates toxins, and the environment around the cell supports the cell’s chemical activities. If the cell is not able to maintain the correct balance of magnesium, potassium and sodium within itself (and also calcium), then potassium and magnesium will be lost, and sodium levels will build up in the cell (processes that utilise calcium, such as muscle contraction and nerve excitability, become overactive -- calcium helps muscles to contract, and magnesium helps them to relax).
Cellular processes reliant on magnesium and potassium, including DNA synthesis, enzyme production (and function), and energy production, become sluggish: the cell becomes stagnant.
At the same time, the environment of the cell changes: pH (acid-alkaline balance), temperature, hydration and light availability. This affects many other cellular activities, and together with the increased toxic load, may lead to chronic inflammation(1) and impaired function, including changes in pH (the body becomes acidic) and the slowing of the metabolic rate: the cell receives less oxygen, gradually reducing the ability to take up oxygen in the lungs and by the blood. Usually, the root cause of many cancers.
Otto Warburg, awarded Nobel prizes for his work on cancer, said that “Cancer has only one prime cause. The prime cause of cancer is the replacement of normal oxygen respiration of body cells by an anaerobic (oxygen-less) cell respiration.”
Magnesium also helps regulate blood sugar levels, promotes normal blood pressure(2), and is known to be involved in protein synthesis(3). Dietary magnesium is absorbed in the small intestines and excreted through the kidneys.
Most of the magnesium in the body is found in the bones (50%) and inside the cells of body tissues and organs. Only a tiny amount of magnesium is normally present in the blood (1%); however, the body works very hard to keep this concentration constant(4). A lack of magnesium can also cause contractions in the arterial walls leading to headaches and migraines.
Looking again at the electrolyte exchange at the cell membrane, if the calcium-magnesium pump is not working efficiently, due to a lack of magnesium, then calcium will build up in the cells and have restricted access back into the blood -- often a leading factor in arthritis. Magnesium actually helps calcium to be absorbed in the first place and stored inside the bones in the form of calcium phosphate crystals. Magnesium also helps calcium to stay in solution in the body; preventing the formation of "stones" (e.g. gallstones).
Neurotransmitters in the brain use magnesium, calcium and zinc to stimulate and regulate electrical activity. Calcium-rich brain cells can fire excessively, which can deplete their energy very quickly and, sometimes, lead to seizures, or even cell death. Magnesium lowers the threshold for those seizures, and so reduces the likelihood that they will happen. Brain cells damaged by toxicity will also fire inappropriately. Chemicals such as food additives, pesticides, herbicides, solvents and cleaning products can (for the most part) cross the blood-brain barrier and poison brain cells, even more so in the case of leaky brain (a process similar to leaky gut. This subject is extensively discussed in my upcoming books). Magnesium can help to protect the brain from the toxic effects of these chemicals.
Magnesium is also required for the production of serotonin, making it a useful nutrient for those with depression, and also for the production of dopamine, low levels of which are associated with Parkinson’s disease.
Fluoride detox side effects
Fluoride detoxification can elicit side effects as the body works to eliminate excess fluoride, a naturally occurring element added to water supplies in several countries, and dental products.
Worldwide, the Irish Republic and Singapore are the only countries implementing mandatory water fluoridation. However, a further 9 countries expose over half their population to fluoridated drinking water:
Australia (80%)
Brunei (95%)
Chile (70%)
Guyana (62%)
Hong Kong (100%)
Israel (70%)
Malaysia (75%)
New Zealand (62%)
United States (64%)
When undergoing fluoride detox, individuals may experience symptoms like headaches, fatigue, muscle and joint pain, gastrointestinal disturbances, and skin issues. The side effects are typically transient and may vary in severity depending on the individual's exposure to fluoride.
One crucial element in fluoride detox is magnesium. Magnesium not only helps support the body's natural detoxification processes but also counteracts some of the potential negative effects of fluoride exposure. It does so by binding to fluoride ions, reducing their bioavailability, and aiding their excretion through the kidneys.
Additionally, magnesium supports overall health by maintaining proper enzyme function, muscle and nerve function, and bone health. To enhance fluoride detoxification and mitigate side effects, ensuring an adequate intake of magnesium through dietary sources or supplements can be a valuable strategy.
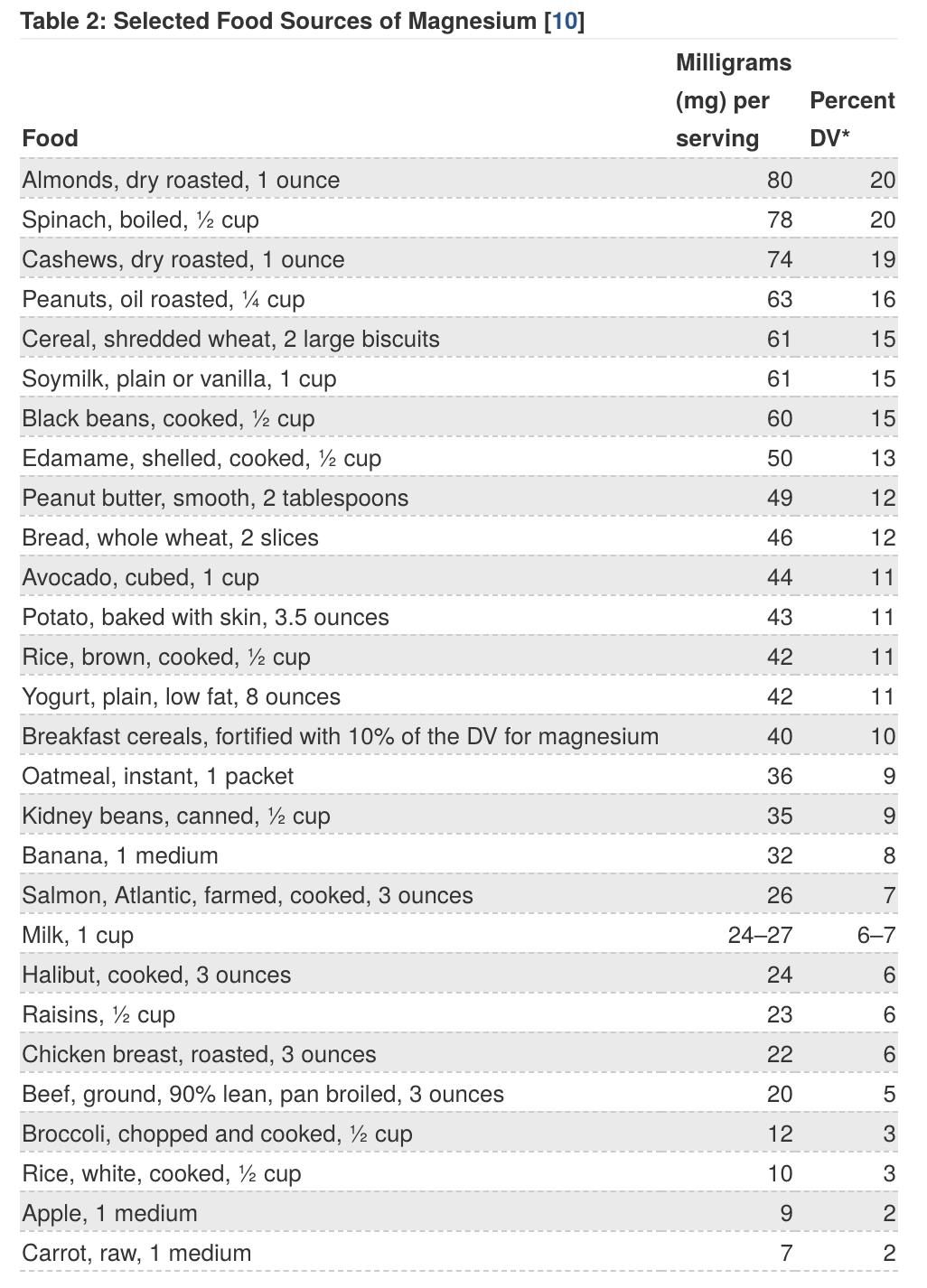
What food contains magnesium?
Shellfish have the highest levels of magnesium.
Eating a wide variety of whole grains, legumes, and vegetables (especially dark-green, leafy vegetables) daily will help provide the recommended intake of magnesium and maintain normal storage. Green vegetables such as spinach, Kale, and other vegetables of the Brassicae family are good sources of magnesium because magnesium is at the core of the chlorophyll molecule. Some legumes (beans and peas), avocado, nuts and seeds, and whole, unrefined grains are also good sources of magnesium.
Avoid processed and refined cereals-based food, including bread and pasta; the magnesium-rich germ and bran have been removed (15g of wheat bran contains 89 mg of magnesium)(5), leaving a product literally minerals-free (and fibre-free), made mainly of carbohydrates (including lots of added sugar, and salt).
Almonds contain as much magnesium as wheat bran, making it an ideal snack (and is also very rich in essential fatty acids). Other nuts such as cashews are also rich in Magnesium.
Beans and rice have lower levels of magnesium (a portion contains between 35–50 mg); however, they are still considered to be a good source(5), as they are able to cover between 10–19% of the Daily Value (the DV for magnesium is 400 milligrams (mg). See table below).
It is important to note that, in general, boiling vegetables and freezing will dramatically reduce their mineral content, including magnesium.
Magnesium deficiency
A healthy digestive system and kidneys can greatly influence the absorption and magnesium retention. Approximately one-third to one-half of dietary magnesium is absorbed into the body. Gastrointestinal disorders (such as Crohn’s disease) impair absorption and can limit the body’s ability to absorb magnesium, deplete the body’s stores of magnesium and, in extreme cases, may result in magnesium deficiency.
Chronic or excessive vomiting and diarrhoea may also result in magnesium depletion(6).
Healthy kidneys are able to limit urinary excretion of magnesium to make up for low dietary intake; however, excessive loss of magnesium can be provoked by some medications (including some antibiotics, Cortisone, insulin, the oral contraceptive pill, cancer-fighting drugs (anti-neoplastic medication), diuretics(7), caffeine and phosphates (also found in fizzy drinks), and can also occur in cases of poorly-controlled diabetes(8) and alcohol abuse(9).
Magnesium supplementation may be indicated when a specific health problem(10) or condition causes an excessive loss of magnesium or limits magnesium absorption. Chronic malabsorptive problems such as Crohn’s disease, gluten-sensitive enteropathy, regional enteritis, and intestinal surgery may generate loss of magnesium through diarrhoea and fat malabsorption(11).
Magnesium absorption decreases with age and renal excretion of magnesium increases, which can be made worse when also taking drugs that interact with magnesium.
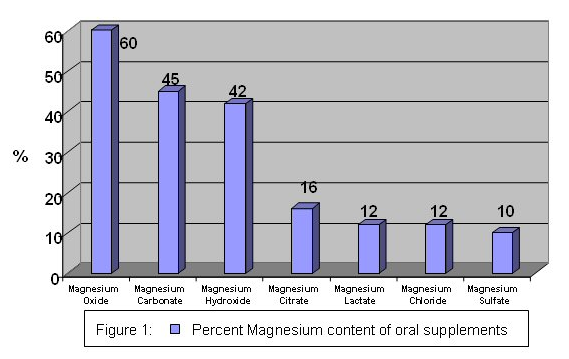
Oral magnesium supplements are a combination of magnesium and another substance, often salts (magnesium oxide, magnesium sulphate, magnesium carbonate, etc.). Elemental magnesium refers to the amount of magnesium in each compound.
The diagram below compares the amount of elemental magnesium in different types of magnesium supplements(12).
The amount of elemental magnesium in a compound and its bioavailability influence the effectiveness of the magnesium supplement.
Bioavailability refers to the amount of magnesium in food, medications, and supplements that is absorbed in the intestines and ultimately available for biological activity in your cells and tissues. It is also important to mention that the outer layer of a Magnesium tablet or capsule that allows it to pass through the stomach and to be dissolved in the small intestine can decrease its bioavailability(13).
The results of a study comparing four forms of magnesium preparations suggested lower bioavailability of magnesium oxide, with significantly higher and equal absorption and bioavailability of magnesium chloride and magnesium lactate(14). This supports the belief that both the magnesium content of a dietary supplement and its bioavailability contribute to its ability to restore deficient levels of magnesium.
For best uptake by the cells, magnesium requires an alkaline environment and good levels of vitamin B6, as well as high levels of stomach acid.
Source: Office of Dietary Supplements website: “Dietary Supplement Fact Sheet: Magnesium.”
*DV = Daily Value. DVs were developed by the FDA to help consumers compare the nutrient contents of products within the context of a total diet. The DV for magnesium is 400 mg for adults and children aged 4 and older. However, the FDA does not require food labels to list magnesium content unless a food has been fortified with this nutrient. Foods providing 20% or more of the DV are considered to be high sources of a nutrient.
The information is provided to demonstrate the variable amount of magnesium in magnesium supplements.
What are the risks of taking magnesium?
Many supplements contain carbonates and oxides, as these are cheaper forms; however, these forms will also reduce stomach acidity and, therefore, how well absorbed the magnesium is. To be assimilated, magnesium requires stomach acid and as such, supplemental magnesium must always be taken with food.
It is the acidity of the stomach that breaks down magnesium so it can be assimilated by the body.
Magnesium chloride is the form that the stomach converts magnesium into.
Side effects: Magnesium supplements can cause nausea, cramps, and diarrhoea (most laxatives contain magnesium sulphate or oxide because it floods the colon with water). Magnesium supplements often cause softening of stool. Overdosing may increase the symptoms dramatically, including low blow pressure, Muscle weakness and fatigue. At very high doses it can be fatal.
Interactions. Magnesium supplements are not recommended for people who take diuretics, heart medicines, or antibiotics.
Risks. People with diabetes, intestinal, kidney or Heart diseases should avoid taking supplementation without the advice of a Doctor or Health practitioner.
Magnesium Sulphate (Epsom salt) is best used externally to relax muscle when poured into a warm bath.
Magnesium is so important to our body that research is constantly trying to understand the mineral's role in our body and health.
Magnesium and Detoxification
Our detoxification processes require energy supplied by the adenosine triphosphate (ATP) molecule. So without enough magnesium, toxicity can build up in our cells and damage their functions further still = poisoning. We are essentially made of approximately 100 trillion cells. If our cells are lacking energy and underperforming, then so are we.
Magnesium is also essential for the production of glutathione (one of the most powerful antioxidant enzymes naturally produced by the body) which plays a pivotal role in cellular and liver detoxification pathways.
In addition, the importance of magnesium to cellular integrity and function described above shows just how crucial magnesium is to general tissue health in the body, including the tissue lining the bowel.
Together with zinc, vitamin C and essential fatty acids, magnesium can help reduce inflammation and repair damage to the inside of the digestive tract, and so help with IBS, Crohn’s disease, colitis and many conditions arising from malabsorption in the gut.
To avoid the occurrence or reoccurrence of bowel disorders, we need to ensure that our intestines have the resources they need to stay healthy, and the correct magnesium:calcium ratio to avoid spasms and help the bowel move in a way that helps prevent stagnation and fermentation.
Phytic acid and lectins found in grains, seeds and legumes, prevent the absorption of magnesium. Soaking and sprouting can help remove phytates and lectins.
(read more: https://www.nutrunity.com/updates/lectins)
Heavy metals compete with magnesium for absorption in the small intestine and brain, and so — as well as depleting magnesium —, heavy metal intake will be reduced if we consume enough magnesium-rich foods and if enough magnesium is absorbed by the small intestine.
Magnesium also plays an important role in detoxifying our cells from heavy metals, not least through the production of detoxifying glutathione. Studies have shown that magnesium, in fact, protects the body from heavy metal effects and damage(15)
As magnesium increases detoxification pathways in the body(16), we need to make sure that the routes of elimination can cope with the extra workload. This may require additional support for the lymphatic system, blood, liver, kidneys, lungs, skin and/or colon.
Always seek advice from your health practitioner. Never try a detoxification program unsupervised, and remember it may get worse before it gets better: also known as a healing crisis. (read previous articles on detoxification)
Chronic stress and anxiety also increase magnesium requirements. So it is vital to address your level of stress, exposure to stressful compounds (EMFs, pollution, etc.) adding extra pressure on the body and systems, and activating the stress response (I urge you to read all articles on the HPA axis and stress on this platform).
References
Mark Sircus tells of a Medical University of South Carolina study of blood inflammation levels, using C-reactive protein as a marker, in 3800 men and women, which showed that those who got consumed than 50% of the RDA for magnesium (310-420mg) were almost 3 times more likely to have dangerously high CRP levels. (“Inflammation and Systemic Stress” 2008)
Wester PO. Magnesium. Am J Clin Nutr 1987;45:1305-12.
Saris NE, Mervaala E, Karppanen H, Khawaja JA, Lewenstam A. Magnesium: an update on physiological, clinical, and analytical aspects. Clinica Chimica Acta 2000;294:1-26.
Institute of Medicine. Food and Nutrition Board. Dietary Reference Intakes: Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride. National Academy Press. Washington, DC, 1999
Rude RK. Magnesium deficiency: A cause of heterogeneous disease in humans. J Bone Miner Res 1998;13:749-58.
U.S. Department of Agriculture, Agricultural Research Service. 2011. USDA National Nutrient Database for Standard Reference, Release 24.
Rude KR. Magnesium metabolism and deficiency. Endocrinol Metab Clin North Am 1993;22:377-95.
Ramsay LE, Yeo WW, Jackson PR. Metabolic effects of diuretics. Cardiology 1994;84 Suppl 2:48-56.
Tosiello L. Hypomagnesemia and diabetes mellitus. A review of clinical implications. Arch Intern Med 1996;156:1143-8.
Elisaf M, Bairaktari E, Kalaitzidis R, Siamopoulos K. Hypomagnesemia in alcoholic patients. Alcohol Clin Exp Res 1998;22:244-6.
Abbott L, Nadler J, Rude RK. Magnesium deficiency in alcoholism: Possible contribution to osteoporosis and cardiovascular disease in alcoholics. Alcohol Clin Exp Res 1994;18:1076-82.
Shils ME. Magnesium. In Modern Nutrition in Health and Disease, 9th Edition. (edited by Shils, ME, Olson, JA, Shike,
M, and Ross, AC.) New York: Lippincott Williams and Wilkins, 1999, p. 169-9
American Diabetes Association. Nutrition recommendations and principles for people with diabetes mellitus. Diabetes Care 1999;22:542-5.
Rude RK and Olerich M. Magnesium deficiency: Possible role in osteoporosis associated with gluten-sensitive enteropathy. Osteoporos Int 1996;6:453-61.
Klasco RK (Ed): USP DI® Drug Information for the Healthcare Professional. Thomson MICROMEDEX, Greenwood Village, Colorado 2003.
Fine KD, Santa Ana CA, Porter JL, Fordtran JS. Intestinal absorption of magnesium from food and supplements. J Clin Invest 1991;88:296-402.
Firoz M and Graber M. Bioavailaility of US commercial magnesium preparation. Magnes Res 2001;14:257-62.
M Speich et al “Correlations between magnesium and heavy metals in blood and sixteen tissues of rabbits.” Magnes Res. Sep 1989
16. by increasing glutathione levels, aiding in the production of ATP and helping to drive electrolyte exchange at the cell membrane
Institute of Medicine (IOM). Food and Nutrition Board. Dietary Reference Intakes: Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride. Washington, DC: National Academy Press, 1997.
U.S. Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 25. Nutrient Data Laboratory Home Page, 2012.
More infos:
S Apte & R Sarangarajan “Cellular Respiration and Carcinogenesis” (Humana Press 2009)
J Anastassapoulos & T Theophanides “Magnesium–DNA interactions and the possible relation of magnesium to carcinogenesis. Irradiation and free radicals“ Oncology Hematology April 2002
Dr Carolyn Dean “The Miracle of Magnesium” Ballantine Books 2003
D Thomas “A study on the mineral depletion of the foods available to us as a nation” 2003Dr Carolyn Dean “The Miracle of Magnesium” Ballantine Books 2003
LG Plaskett, “The Truth About Mineral Supplementation” Nutrigold 2000