The Hidden Engine of Your Body: How Mitochondria Drive Energy, Health, and Performance
Mitochondria are often described as the “powerhouses” of our cells, but this phrase only scratches the surface of their vital role in human health. These microscopic organelles are responsible for producing ATP (adenosine triphosphate), the universal energy currency that powers every biological process, from muscle contraction and brain function to cell repair, hormone synthesis and detoxification.
Understanding how ATP is made, why it’s so essential, and how we can optimise mitochondrial health is key to unlocking better energy, resilience, and even longevity.
What Is ATP and Why Is It So Important?
ATP is a small but mighty molecule produced in every body cell by mitochondria. Structurally, it consists of adenine (a nitrogenous base), ribose (a sugar), and three phosphate groups. The energy stored in ATP is held in the bonds between these phosphate groups, particularly the last bond. When the body needs energy, an enzyme breaks this bond, releasing energy that powers countless cellular functions.
Key roles of ATP include:
Muscle contraction: Every movement, from blinking to running, relies on ATP.
Brain function: Neurones use ATP to transmit signals and support cognitive processes.
Cell growth and repair: ATP fuels the synthesis of proteins, membranes, and DNA.
Detoxification: The breakdown of toxins and metabolic waste depends on ATP-driven reactions.
Without ATP, life as we know it would grind to a halt. The body’s demand for ATP is so high that each molecule is recycled thousands of times daily.
How Do Mitochondria Produce ATP?
— The Basics of Cellular Respiration
Mitochondria generate ATP through cellular respiration, which transforms the energy stored in food into usable fuel for the body. This multi-step process includes:
Glycolysis: Glucose (from carbohydrates) is broken down in the cytosol (space inside a cell) into pyruvate, producing a small amount of ATP and NADH.
Citric Acid Cycle (Krebs Cycle): Pyruvate enters the mitochondrial matrix and is converted to Acetyl-CoA, which is further oxidised, generating NADH and FADH₂.
Electron Transport Chain (ETC): NADH and FADH₂ donate electrons to the ETC, a series of protein complexes in the inner mitochondrial membrane. This drives the pumping of protons (H⁺) across the membrane, creating an electrochemical gradient.
ATP Synthase: Protons flow back into the mitochondrial matrix through ATP synthase, a molecular machine that uses this flow to generate ATP from ADP and phosphate.
This process is highly efficient: one glucose molecule can yield up to 30–32 molecules of ATP when oxygen is plentiful. In contrast, glycolysis alone (without oxygen) produces just 2 ATP per glucose, highlighting the critical importance of mitochondria and oxygen for optimal energy production.
Basic overview of cell, processes of ATP production from macronutrients (proteins, carbs and fatty acids), including mitochondrial energy production via the Krebs cycle and the ETC (also known as phosphorylation) in aerobic and anaerobic cellular environments. Straight lines represent catabolic metabolism and doted lines anabolic metabolism. Diagram is extracted from “Detox before Energise.” (p. 301). Copyright: Olivier Sanchez, Nutrunity Publishing.
How Does ATP Work?
ATP (adenosine triphosphate) is the body’s universal energy carrier; without it, life simply couldn’t exist. Every cell relies on ATP as its immediate energy source to power vital functions like muscle contraction, nerve signal transmission, tissue repair, and growth.
Chemically, ATP is made up of three parts: adenine (a nitrogen base), ribose (a sugar), and a chain of three phosphate groups. The energy that fuels our cells is stored in the bonds between these phosphate groups, especially the last one. When the body needs energy, an enzyme breaks this final phosphate bond, releasing a burst of energy that cells use for everything from moving muscles to sending nerve signals.
This process transforms ATP into ADP (adenosine diphosphate) and a free phosphate group. Importantly, ATP isn’t just used up and discarded; it’s constantly recycled within our cells, ensuring a steady energy supply for all bodily processes. This continuous cycle keeps our metabolism running and supports everything from daily activities to intense exercise.
Beyond Energy: The Multifaceted Role of Mitochondria
While ATP production is their most famous function, mitochondria do much more:
Biosynthesis: Mitochondria provide building blocks for macromolecules like lipids, proteins, and nucleotides, supporting cell growth and repair (see illustration above).
Redox Balance: They help regulate the cell’s redox state, managing reactive oxygen species (ROS — free radicals) and maintaining antioxidant defences.
Waste Management: Mitochondria process metabolic by-products and play an essential role in detoxification.
Cell Signalling: ATP and other mitochondrial products act as signals, influencing everything from hormone release to immune responses.
Apoptosis: Mitochondria help control programmed cell death and are vital for tissue health and cancer prevention.
Mitochondria and Hormone Synthesis
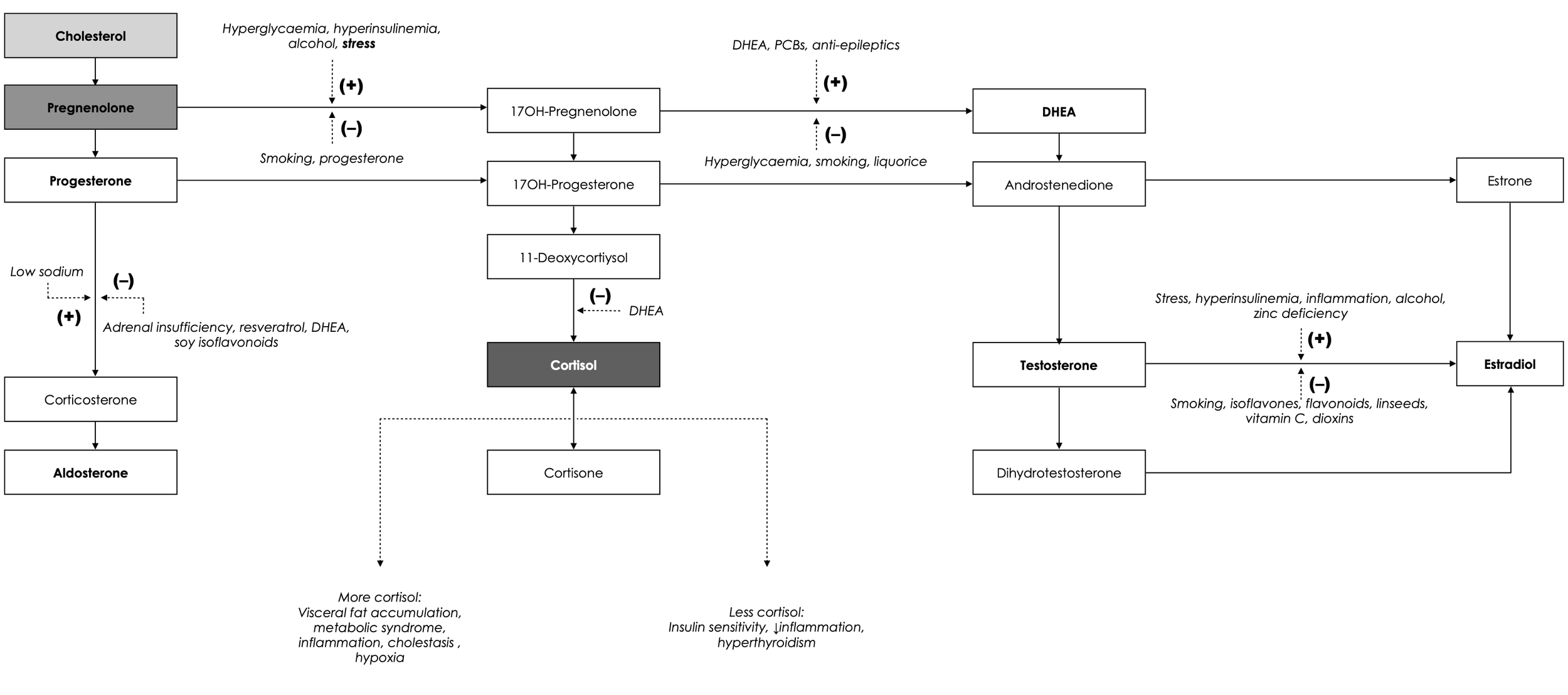
Mitochondria are fundamental to synthesising steroid hormones, including cortisol, aldosterone, oestrogen, progesterone, and testosterone. The first and critical step in steroid hormone biosynthesis occurs within the mitochondria: converting cholesterol to pregnenolone by the enzyme P450scc (cholesterol side-chain cleavage enzyme). This process occurs in steroid-producing cells of the adrenal glands, gonads, placenta, and even the brain, where mitochondria house P450scc and other key enzymes involved in steroidogenesis.
The steroidogenic acute regulatory protein (StAR) and a complex of associated proteins on the outer mitochondrial membrane tightly regulate cholesterol transport into mitochondria. Once inside, cholesterol is converted to pregnenolone, further processed into various steroid hormones by additional enzymes.
Hormone Secretion and Poor Mitochondrial Function
Proper mitochondrial function is vital for hormone synthesis and the regulation and secretion of hormones. When mitochondrial function is compromised, several problems can arise:
Reduced Hormone Production: Deficiencies in mitochondrial enzymes or impaired mitochondrial dynamics (such as poor fusion) can directly reduce the synthesis of steroid hormones, leading to hormonal imbalances and related health issues.
Disrupted Hormonal Regulation: Mitochondria are both regulated by and regulators of hormones. For example, sex steroid hormones like oestrogen can modulate mitochondrial function, and in turn, mitochondria influence the availability and action of these hormones. Poor mitochondrial health can therefore disrupt this feedback loop, further impairing hormonal balance.
Impaired Hormone Secretion: ATP produced by mitochondria is essential for the energy-intensive processes involved in hormone secretion. If mitochondrial ATP production drops, cells may not secrete hormones efficiently, impacting everything from stress response (cortisol) to reproductive health (oestrogen, testosterone).
Broader Systemic Effects: Because steroid hormones regulate vital processes such as metabolism, stress response, immune function, and reproduction, mitochondrial dysfunction can contribute to various disorders, including adrenal insufficiency, infertility, and neuroendocrine diseases.
Cholesterol, the precursor to all steroid (sex) hormones: Steroidogenesis. Pathways antagonists and agonists. Copyright: Olivier Sanchez.
Why Is Mitochondrial Health So Critical?
When mitochondria function well, we feel energetic, focused, and resilient. When they falter, the effects are wide-ranging: fatigue, brain fog, muscle weakness, poor recovery, hormone imbalances and even chronic diseases. Mitochondrial dysfunction is implicated in conditions such as diabetes, neurodegenerative diseases, cardiovascular disease, and accelerated ageing.
Maintaining a high ATP:ADP ratio is essential:
Cells keep ATP levels about 10 times higher than ADP to ensure that energy-requiring reactions can proceed efficiently. If mitochondrial activity is blocked (for example, by toxins like cyanide or heavy metals), ATP levels drop, and vital cellular functions quickly shut down.
How Can You Optimise ATP Production and Mitochondrial Health?
1. Nutrition: Feed Your Powerhouses
Magnesium: Essential for over 300 enzymatic reactions, including those that generate ATP. Foods rich in magnesium include dark leafy greens, nuts, seeds, and avocados.
B Vitamins: Especially B1 (thiamine), B2 (riboflavin), B3 (niacin), and B5 (pantothenic acid). These act as cofactors in the energy metabolism and detoxification pathways. Sources include whole grains, eggs, legumes, and lean meats.
Alpha-Lipoic Acid: A powerful antioxidant and cofactor in the citric acid (Krebs) cycle, found in spinach, broccoli, and organ meats.
Coenzyme Q10 (CoQ10): Vital for electron transport in the ETC. Rich sources are fatty fish and organ meats.
Antioxidants & Phytochemicals: Brightly coloured fruits and vegetables (berries, peppers, kale, turmeric, ginger) protect mitochondria from oxidative stress and support their function. Glutathione and SOD (superoxide dismutase), two major antioxidant enzymes produced by the body are the primary protectors of mitochondria against ROS. Supplementing with these two compounds is not recommended because they are poorly absorbed, and (medical-grade) therapeutic supplements are very costly (e.g., liposomal reduced glutathione). They may, however, be considered if recommended by your practitioner.
2. Exercise: Move to Multiply Mitochondria
Regular physical activity, especially aerobic exercise and high-intensity interval training (HIIT), stimulates the production of new mitochondria (mitochondrial biogenesis) and enhances efficiency. Exercise also helps regulate mitochondrial quality by promoting the removal of damaged organelles and increasing ATP production capacity.
3. Sleep: Recharge Your Cellular Batteries
Quality sleep is essential for mitochondrial repair and renewal. During deep sleep, the body performs maintenance checks on mitochondria, ensuring they’re ready to meet energy demands the next day.. Chronic sleep deprivation impairs mitochondrial function and leads to decreased ATP production, brain fog, low mood and fatigue.
4. Stress Management: Protect Against Burnout
Chronic stress increases oxidative damage and impairs mitochondrial function. Mindfulness, meditation, and relaxation techniques can lower stress hormones and help maintain mitochondrial health.
5. Intermittent Fasting: Increase Mitochondrial Efficiency
Intermittent fasting, cycling between periods of eating and extended fasting, has been shown to promote autophagy, the process by which damaged mitochondria are cleared out and replaced by new ones. This supports long-term mitochondrial health and optimises ATP production.
6. Hydration: Essential for Cellular Function
Adequate hydration is vital for all cellular processes, including ATP production. Even mild dehydration can impair mitochondrial efficiency and reduce energy levels.
The Evolutionary Story: Mitochondria’s Bacterial Origins
The story of mitochondria is a fascinating chapter in evolutionary biology. According to the endosymbiotic theory, mitochondria originated as free-living bacteria engulfed by ancestral eukaryotic cells over a billion years ago. Instead of being digested, these bacteria formed a symbiotic relationship with their host, exchanging protection and nutrients for the ability to produce energy more efficiently.
Key evidence for this theory includes:
Mitochondria have their own DNA (mtDNA), distinct from nuclear (our human) DNA.
They possess a double membrane, similar to bacteria.
Mitochondrial ribosomes (the organelles responsible for energy production) resemble those of bacteria, not eukaryotic cells.
Certain antibiotics that target bacteria also affect mitochondrial protein synthesis.
This ancient partnership allowed complex multicellular life to evolve, as cells could now generate enough energy to support larger, more intricate organisms.
Signs of Mitochondrial Dysfunction
How do you know if your mitochondria need support?
Common signs include:
Persistent fatigue or low energy, and difficulties waking up in the morning (also linked to cortisol)
Muscle weakness or slow recovery after exercise
Brain fog, memory issues, or poor concentration
Poor tolerance to exercise or physical activity
Frequent illnesses or slow healing
Early signs of ageing (wrinkles, hair loss, loss of vitality)
If you experience several of these symptoms, optimising your mitochondrial health may help restore your energy and resilience.
Mitochondria and hormones
When mitochondrial function is compromised, several problems can arise:
Reduced Hormone Production: Deficiencies in mitochondrial enzymes or impaired mitochondrial dynamics can directly reduce the synthesis of steroid hormones, leading to hormonal imbalances and related health issues.
Disrupted Hormonal Regulation: Mitochondria are both regulated by and regulators of hormones. For example, sex steroid hormones like estrogen can modulate mitochondrial function, and in turn, mitochondria influence the availability and action of these hormones. Poor mitochondrial health can therefore disrupt this feedback loop, further impairing hormonal balance.
Impaired Hormone Secretion: ATP produced by mitochondria is essential for the energy-intensive processes involved in hormone secretion. If mitochondrial ATP production drops, cells may not secrete hormones efficiently, impacting everything from stress response (cortisol) to reproductive health (oestrogen, testosterone).
Broader Systemic Effects: Because steroid hormones regulate vital processes such as metabolism, stress response, immune function, and reproduction, mitochondrial dysfunction can contribute to a wide range of disorders, including adrenal insufficiency, infertility, and neuroendocrine diseases.
“Mitochondrial dysfunction can contribute to a wide range of disorders, including adrenal insufficiency, infertility, and neuroendocrine diseases.”
Mitochondria and Neurodegeneration
Mitochondrial dysfunction is increasingly recognised as a central player in cognitive impairment and neuropsychiatric disorders, with emerging research highlighting its role in brain metabolism and energy dynamics.
Emerging Roles of Brain Metabolism in Cognitive Impairment and Neuropsychiatric Disorders
— Mitochondria as Energy Hubs in Cognitive Function
The brain consumes around 20% of the body’s oxygen and glucose, relying heavily on mitochondrial ATP production to sustain high-energy demands for neuronal signalling, neurogenesis, and synaptic plasticity. Disruptions in mitochondrial respiration or calcium buffering impair memory and learning. For example, AIF-deficient models show depleted neural stem cell pools and defective adult neurogenesis, directly linking mitochondrial dysfunction to cognitive decline.
Note: AIF (Apoptosis-Inducing Factor) is a mitochondrial protein with dual roles in regulating cell death and maintaining mitochondrial function and cellular energy metabolism.
— Neuropsychiatric Symptoms and Oxidative Stress
Mitochondrial abnormalities drive neuropsychiatric disorders such as schizophrenia and bipolar disorder through energy perturbations and reactive oxygen species (ROS) accumulation. ROS overproduction damages neuronal membranes, disrupts neurotransmitter systems (e.g., glutamate), and alters self-referential thoughts and emotional processing.
— Mitochondrial Allostatic Load and Stress Adaptation
Chronic stress triggers a “mitochondrial allostatic load,” where prolonged glucocorticoid and catecholamine signalling disrupts mitochondrial dynamics, reduces ATP synthesis, and promotes inflammation. This metabolic strain exacerbates cognitive deficits in conditions like Alzheimer’s disease (AD), where mitochondrial dysfunction accelerates plaque deposition.
— Therapeutic Implications
Targeting mitochondrial pathways offers promise for treating cognitive and psychiatric symptoms. Natural compounds like resveratrol improve mitochondrial biogenesis and reduce oxidative stress. Emerging strategies also focus on modulating stress-induced mitochondrial adaptations to prevent neurodegeneration.
This synthesis places mitochondria as metabolic gatekeepers in brain health, bridging energy production, stress responses, neuroinflammation and neurodegeneration.
How to Support Your Mitochondria Every Day
Eat a colourful, varied, nutrient-dense diet rich in antioxidants, B vitamins, magnesium, and healthy fats.
Exercise regularly, combining aerobic activities with some high-intensity intervals.
Prioritise sleep: aim for 7.5–9 hours per night (sleep uses cycles of 90 minutes).
Manage stress with mindfulness, breathing exercises, or journaling.
Stay hydrated throughout the day.
Consider intermittent fasting (with professional guidance) to promote cellular renewal.
Limit exposure to toxins (like cigarette smoke, excess alcohol, and environmental pollutants, including heavy metals) that can damage mitochondria.
Review medications with your healthcare provider, as some drugs can impair mitochondrial function.
Power Up Your Life by Supporting Your Cellular Engines
Mitochondria are the hidden engines that power every aspect of your health, vitality, and performance. By understanding how these remarkable organelles work and taking steps to support their function through nutrition, exercise, sleep, and stress management, you can unlock more energy, sharper thinking, and greater resilience at every stage of life.
Whether you’re an athlete seeking peak performance, a busy professional battling fatigue, or simply looking to age well, investing in your mitochondrial health is one of the most effective ways to boost your energy from the inside out.
References
Alshial, EE. Abdulghaney, MI. Wadan, AS. et al. (2023). Mitochondrial dysfunction and neurological disorders: A narrative review and treatment overview. Life Science. 334, 122257. doi:10.1016/j.lfs.2023.122257
Brand, MD. Orr, AL. Perevoshchikova, IV. et al. (2023). The role of mitochondrial function and cellular bioenergetics in ageing and disease. British Journal of Dermatology. 169(Suppl. 2), pp. 1-8. doi:10.1111/bjd.12208
Casanova, A. Wevers, A. Navarro-Ledesma, S. et al. (2023). Mitochondria: It is all about energy. Frontiers in Physiology. 14, 1114231. doi:10.3389/fphys.2023.1114231
Clemente-Suárez, VJ. Redondo-Flórez, L. Beltrán-Velasco, AI. et al. (2023). Mitochondria and brain disease: A comprehensive review of pathological mechanisms and therapeutic opportunities. Biomedicines. 11(9), 2488. doi:10.3390/biomedicines11092488
Dunn, J. Grider, MH. (2025). Physiology, Adenosine Triphosphate. In: StatPearls. Treasure Island (FL): StatPearls Publishing. [Available from: https://www.ncbi.nlm.nih.gov/books/NBK553175]
Hatefi, Y. (1985). The mitochondrial electron transport and oxidative phosphorylation system. Annual Review of Biochemistry. 54, pp. 1015-1069. doi:10.1146/annurev.bi.54.070185.005055
Javadov, S. Kozlov, AV. Camara, AKS. (2020). Mitochondria in health and diseases. Cells. 9(5), 1177.
Meng, K. Jia, H. Hou, X. et al. (2025). Mitochondrial dysfunction in neurodegenerative diseases: Mechanisms and corresponding therapeutic strategies. Biomedicines. 13(2), 327. doi:10.3390/biomedicines13020327
Martin, WF. Garg, S. Zimorski, V. (2025) Endosymbiotic theories for eukaryote origin. Philosophical Transactions of the Royal Society B. 370(1678), 20140330.
Morella, IM. Brambilla, R. Morè, L. (2022). Emerging roles of brain metabolism in cognitive impairment and neuropsychiatric disorders. Neuroscience & Biobehavioral Reviews. 142, 104892. doi:10.1016/j.neubiorev.2022.104892
Norat, P. Soldozy, S. Sokolowski, JD. et al. (2020). Mitochondrial dysfunction in neurological disorders: Exploring mitochondrial transplantation. npj Regenerative Medicine. 5, 22. doi:10.1038/s41536-020-00107-x
Nunnari, J. Suomalainen, A. (2012). Mitochondria: in sickness and in health. Cell. 148(6), pp. 1145-1159. doi:10.1016/j.cell.2012.02.035
Orrenius, S. (2007). Reactive oxygen species in mitochondria-mediated cell death. Drug Metabolism Reviews. 39(2-3), pp. 443-55. doi:10.1080/03602530701468516
Orrenius, S. Gogvadze, V. Zhivotovsky, B. (2007). Mitochondrial oxidative stress: Implications for cell death. Annual Reviews of Pharmacology and Toxicology. 47, pp. 143-183. doi:10.1146/annurev.pharmtox.47.120505.105122
Ott, M. Gogvadze, V. Orrenius, S. et al. (2007). Mitochondria, oxidative stress and cell death. Apoptosis. 12(5), pp. 913-922. doi:10.1007/s10495-007-0756-2
Rossmann, MP. Dubois, SM. Agarwal, S. et al. (2021). Mitochondrial function in development and disease. Disease Models & Mechanisms. 14(6), dmm048912.
San-Millán, I. (2023). The key role of mitochondrial function in health and disease. Antioxidants (Basel). 12(4), 782.
Spinelli, JB. Haigis, MC. (2018). The multifaceted contributions of mitochondria to cellular metabolism. Nature Cell Biology. 20(7), pp. 745-754.
Teixeira, J. Chavarria, D. Borges, F. et al. (2019). Dietary polyphenols and mitochondrial function: Role in health and disease. Current Medicinal Chemistry. 26(19), pp. 3376-3406.
Waseem, R. Shamsi, A. Kazim, SN. et al. (2021). An insight into mitochondrial dysfunction and its implications in neurological diseases. Current Drug Targets. 22(14), pp. 1585-1595. doi:10.2174/1389450121999201230204050
Zong, Y. Li, H. Liao, P. et al. (2024). Mitochondrial dysfunction: Mechanisms and advances in therapy. Signal Transduction and Target Therapy. 9, 124. doi:10.1038/s41392-024-01839-8