Strategies for Heavy Metal Detoxification: Systemic Approaches
Heavy metal toxicity represents a pervasive public health challenge in industrialised societies, with mercury, lead, cadmium, chromium, and arsenic inducing systemic harm through oxidative stress, enzymatic disruption, and DNA damage.
Recent research reveals that effective detoxification requires a dual approach:
1) reducing exposure through environmental and
2) dietary modifications while enhancing the body’s innate elimination pathways via targeted nutritional interventions, lifestyle adaptations, and evidence-based supplementation.
I reviewed the latest research to provide 12 strategies that integrate findings from toxicology, nutritional science, and clinical studies to provide a roadmap for mitigating heavy metal toxicity.
Mechanisms of Heavy Metal Toxicity and Detoxification Pathways
— Bioaccumulation and Organ-Specific Damage
Heavy metals disrupt cellular homeostasis through three primary mechanisms: reactive oxygen species (ROS — free radicals) production, competitive inhibition of essential minerals, and direct binding to sulfhydryl groups (also called “thiol groups,” consisting of a sulfur atom with two lone pairs, bonded to hydrogen) in proteins and enzymes.
Mercury and aluminium preferentially accumulate in neural tissues, impairing glutathione peroxidase activity and inducing neuroinflammation, while cadmium displaces calcium in bone tissue, accelerating osteoporotic degeneration.
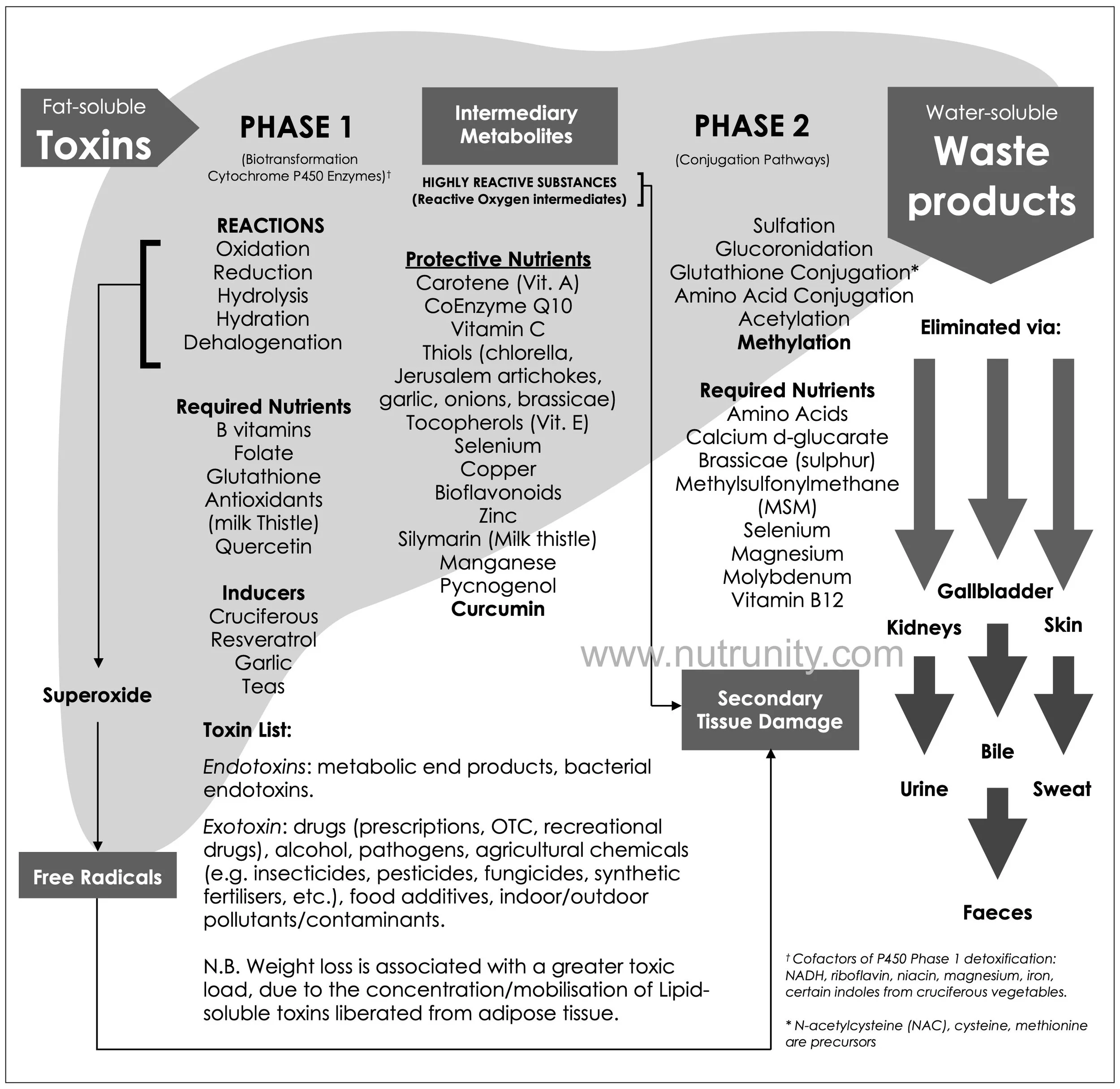
The liver’s phase I and II detoxification pathways — particularly cytochrome P450 enzymes and glutathione conjugation — serve as the frontline defence, converting lipid-soluble toxins into water-soluble metabolites for renal or biliary excretion. Chronic exposure overwhelms these systems, necessitating proactive interventions to reduce toxic loads.
Review of liver detoxification pathways and required nutrients and cofactors.
If you need to review the basics of liver detoxification pathways, click the button below.
Reducing Environmental and Dietary Exposure
— Drinking Water
Municipal water systems in regions with pre-1970s infrastructure remain vulnerable to lead leaching from deteriorating pipelines, solder joints, and brass fittings containing up to 8% lead content. In Scotland alone, 3–5% of private water supplies exceed the 10 µg/L lead threshold due to legacy plumbing components, with even short lead pipe sections (<1 metre) capable of contaminating entire systems.
Industrial groundwater sources face compounded risks, with cadmium and arsenic infiltrating aquifers through mining runoff or improper waste disposal — a concern in former mining regions like Cornwall and South Wales. Many parts of the world face the same problems.
Point-of-use reverse osmosis (RO) systems employ semipermeable membranes, physically excluding 90–99% of dissolved lead, cadmium, and hexavalent chromium. Independent testing demonstrates consistent removal rates of 98.7% for lead (from 15 µg/L to 0.2 µg/L) and 94.5% for arsenic in simulated contaminated water. However, membrane efficiency depends on optimal pressure (50–70 psi) and regular maintenance — neglected pre-filters allowing sediment buildup can reduce metal rejection by 12–18% within six months.
Whole-house RO systems with remineralisation cartridges address both consumption and dermal exposure risks during bathing, where lead absorption through the skin accounts for 6–8% of total uptake. It is also important to flush stagnant water (>15 days - e.g., after a trip away), as heavy metals may accumulate in the pipes. Run tap water for 2 -5 minutes, depending on pressure. This is also important to flush legionella, a bacteria that thrives in humid and warm habitats like taps.
— Seafood Consumption
Methylmercury (MeHg) undergoes exponential bioaccumulation across aquatic food chains, with concentrations increasing by 3–10 orders of magnitude from phytoplankton to apex predators.
Zooplankton grazing on mercury-contaminated algae achieve tissue concentrations 200–500x higher than surrounding waters, initiating a trophic cascade. Small planktivorous fish like anchovies bioaccumulate MeHg at 0.09–0.15 ppm, while large predators like swordfish exceed 0.95 ppm — surpassing the US EPA’s 0.3 ppm safety threshold by 217%. This biomagnification occurs because MeHg’s strong affinity for thiol groups in proteins enables 95–99% retention efficiency at each trophic transfer, unlike organic compounds metabolised by marine organisms.
N.B. Industrialised coastal regions show 4–7x higher mercury concentrations in equivalent species than open ocean populations.
— Ultra-processed Manufactured Foods (UPFs)
Industrial food processing introduces heavy metals through equipment (chromium in stainless steel), additives (aluminium-based baking powders, aluminium equipment and utensils), and agricultural contaminants.
A 2023 cohort study associated ultra-processed food consumption with 23% higher urinary cadmium levels, likely due to phosphate fertilisers and contaminated soils. Emphasising whole, organic produce—particularly those tested for heavy metals by third-party certifiers—can help reduce exposure. This is also pertinent for all teas, particularly tea bags, because they are filled with loose-leaf tea, unwanted leftovers, waste, rodent faeces, insects, and dust.
— Rice and Other Foods
Arsenic contamination in food, particularly rice, has emerged as a significant concern due to its potential health risks. Unlike other cereals, rice accumulates arsenic at concentrations 10–20 times higher, primarily because of its growth in flooded conditions, which increases arsenic solubility in soil and uptake by the plant from excessive use of synthetic (highly toxic petroleum-based) fertilisers.
Arsenic enters the plant through silicon transporters that mistakenly absorb arsenite (a toxic form of arsenic) due to its chemical similarity to silicic acid. This mechanism results in rice accumulating 10 times more arsenic than wheat or barley.
Brown Rice: Contains higher arsenic levels because the bran layer (where arsenic concentrates) is retained during processing. On average, brown rice has 80% more inorganic arsenic than white rice.
White Rice: While lower in arsenic, it loses 75–90% of its nutrients during milling. Basmati and jasmine rice typically contain less arsenic than other types due to their growth conditions
Rice Products: Items like rice cakes, crackers, and rice milk often contain excessive arsenic levels (exceeding regulatory limits for drinking water), and must be avoided.
Other foods like teas may also contribute to dietary arsenic exposure. Tea plants can absorb heavy metals from contaminated soils, particularly lead, cadmium, and fluoride. While tea generally contains less arsenic than rice, excessive consumption of low-quality teas grown in polluted regions may contribute to heavy metal exposure. Opt for organic teas from trusted sources that test for contaminants.
Be aware of non-organic and organic brassicas, known as hyperaccumulators like tea plants — they can absorb high concentrations of heavy metals. To minimise exposure, continue reading to tips and recommendations below.
Health Risks Associated with Arsenic Exposure
Toxicity and Long-Term Effects
Inorganic arsenic is classified as a Group 1 carcinogen by the International Agency for Research on Cancer (IARC). Chronic exposure has been linked to:
Cancer: Increased risk of lung, bladder, and skin cancers.
Neurological Impacts: Cognitive deficits in children exposed prenatally or during early life.
Cardiovascular Disease: Elevated risks due to oxidative stress and endothelial dysfunction.
Vulnerable Populations
Infants and children are particularly at risk because their smaller bodies magnify exposure compared to adults. A single cup of white rice daily can increase arsenic exposure by 65%, posing a disproportionate risk for developing health conditions later in life.
Mitigating Arsenic Exposure from Rice
— Cooking Methods to Reduce Arsenic
Rinsing Before Cooking
Washing rice thoroughly before cooking can reduce surface-bound arsenic by 10–28%. However, this method alone is insufficient for significant reduction.
Soaking
Soaking rice overnight or for 24 hours can help remove high levels of arsenic (keep in fridge). Rinse thoroughly and boil with 3-4 times its volume in water.
Parboiling
Developed by researchers at the University of Sheffield, this method involves parboiling rice before cooking it with fresh water. It removes up to 74% of arsenic in white rice and 50% in brown rice while preserving essential nutrients like zinc and iron.
Excess Water Cooking
Cooking rice with a high water-to-rice ratio (e.g., 3:1-6:1) and draining the excess water reduces arsenic content by 40–60%, though it also leaches out micronutrients.
Seaweed
Adding a small piece of wakame or kumbu when cooking rice, grains, beans and pulses can help trap arsenic (but also so-called anti-nutrients like lectins and phytates). Certain seaweeds (e.g., hijiki) contain high inorganic arsenic levels due to marine bioaccumulation. Avoid hijiki if concerned about heavy metals.
Dietary Diversification as a Protective Strategy
Alternatives to Rice
Quinoa: A seed containing significantly lower levels of arsenic than rice — up to 10 times less toxic arsenic on average.
Millet and Barley: These grains accumulate minimal amounts of heavy metals due to different root absorption mechanisms.
Legumes: Beans and lentils offer low-arsenic carbohydrate alternatives rich in fibre and protein.
Rotating Food Sources
Reducing reliance on rice as a dietary staple limits cumulative exposure. For example, alternating between quinoa, millet, and potatoes can significantly lower overall dietary arsenic intake without compromising nutritional diversity
Heavy Metal Elimination
Liposomal Glutathione Supplementation
Glutathione structure enables direct conjugation with mercury, cadmium, and lead, forming easily excretable complexes. Oral administration typically achieves poor bioavailability due to gastric degradation, but liposomal encapsulation enhances hepatic delivery by 150–200% in clinical trials.
Adjuvant nutrients like milk thistle (silymarin) upregulate glutathione synthesis by activating Nrf2, while trimethylglycine donates methyl groups critical for phase II detoxification. (refer to the above diagram)
Essential Mineral Repletion
Calcium and iron compete with lead and cadmium for intestinal absorption via divalent metal transporter 1 (DMT1). A 2014 randomised trial demonstrated that iron-deficient adults absorbed 45% more dietary cadmium than replete subjects.
Zinc supplementation at 15–30 mg/day displaces cadmium from receptor sites, while selenium’s incorporation into glutathione peroxidase enhances antioxidant capacity against arsenic-induced oxidative stress.
— Phytonutrient
Brassicas supply sulforaphane and indole-3-carbinol, activating glutathione S-transferase and NAD(P)H quinone oxidoreductase 1 — key enzymes in heavy metal conjugation. Curcumin from turmeric chelates lead and cadmium while inhibiting nuclear factor kappa-B (NF-κB)-mediated inflammation in hepatic Kupffer cells.
Marine algae-derived alginates form non-absorbable complexes with heavy metals in the intestinal lumen, reducing systemic absorption by 40–60% in human trials.
Pair seafood meals with:
Selenium-rich foods: Brazil nuts (2.5 µg/g) counteract mercury toxicity through Hg-Se protein complex formation
Dietary fibres: Psyllium husk reduces enterohepatic recirculation by 38% via mercury-binding in the colon
Chlorella and Spirulina*
Chlorella and Spirulina
* Chlorella and spirulina exert protective effects against dietary heavy metals through three primary mechanisms:
Mucopolysaccharide and Phytochelatin Complexation
Chlorella’s cysteine-rich cell wall mucopolysaccharides form insoluble complexes with methylmercury, reducing intestinal absorption by 38–42% in human trials. Spirulina’s C-phycocyanin and allophycocyanin proteins bind cadmium and lead via sulfhydryl groups, achieving 73% and 61% adsorption rates in vitro, respectively.Enterohepatic Recirruption Disruption
Both algae inhibit mercury reabsorption from bile, enhancing faecal excretion. A 2024 study demonstrated that 5g spirulina with mackerel meals reduced urinary mercury by 29% compared to controls.Competitive Mineral Absorption
Spirulina’s high iron content (15.6 mg/10g) competes with lead for DMT1 uptake, decreasing blood lead levels by 18% in regular seafood consumers
N.B. For extra activity, take chlorella and spirulina before a seafood meal to saturate the intestines and catch biliary excreated metals 20 minutes after the meal.
Lifestyle Modifications for Sustained Detoxification
— Time-Restricted Feeding
A 2021 clinical study on 10-day fasting demonstrated 14.6% and 18.3% reductions in urinary nickel and lead, respectively, likely through autophagy-mediated release of stored metals. Daily fasting windows for 14–16 hours enhance ketone production, which crosses the blood-brain barrier to support neuronal detoxification pathways.
Post-fast reintroduction of foods should emphasise metal-chelating agents like coriander and chlorella to prevent redistribution from adipose tissue.
— Infrared Sauna Therapy
Combining sauna-induced sweating (excreting cadmium and mercury) with electrolyte-balanced hydration may complement renal excretion. Further research is needed to quantify efficacy relative to other modalities.
— Magnesium and Detox
Magnesium is a powerhouse supporting the body’s detoxification processes:
Enhancing cellular energy production:
Magnesium is essential for ATP production — the energy currency of the cell. The liver, the body’s primary detoxification organ, requires large amounts of energy to perform its detox duties, such as converting toxins into water-soluble substances that can be excreted. Magnesium ensures that ATP is available to power these processes, enabling the liver to eliminate heavy metals.
Maintaining glutathione levels:
Glutathione is the body’s primary antioxidant, playing a key role in detoxifying harmful substances. Magnesium is needed for glutathione-producing enzymes, helping the body neutralise free radicals generated by heavy metal exposure. Low magnesium levels result in diminished glutathione production, compromising your body’s ability to detoxify heavy metals effectively.
Reducing inflammation and oxidative stress:
Heavy metals cause oxidative stress, which leads to inflammation. Magnesium reduces the production of pro-inflammatory compounds and stabilises free radicals, preventing them from causing further cellular damage. This anti-inflammatory effect helps mitigate the damage caused by toxins during detoxification, allowing the body to heal and repair.
Chelating heavy metals:
Magnesium itself binds to heavy metals such as lead and mercury through a process called chelation. This binding process allows the metals to be safely excreted from the body, primarily through the kidneys and urine, thereby reducing the overall toxic burden on your system.
— Probiotic Supplementation
Lactobacillus and Bifidobacterium strains express surface proteins that bind lead and cadmium in the gut lumen. A 6-month intervention with multispecies probiotics reduced urinary arsenic by 32% in endemic Bangladeshi populations by competing with arsenic for aquaglyceroporin uptake.
— Chelation therapy
Chelation therapies face limitations in addressing neurotoxic metals like mercury and lead due to poor blood-brain barrier permeability. Emerging evidence highlights the role of endogenous antioxidants like glutathione in mitigating neuronal oxidative stress, with liposomal delivery systems enhancing central nervous system bioavailability. Simultaneously, dietary fibres and clinoptilolite clays show promise in binding heavy metals within the gastrointestinal lumen, preventing enterohepatic recirculation.
Integrating Science into Daily Practice
Heavy metal detoxification transcends simplistic “cleanse” protocols, demanding sustained dietary vigilance, targeted nutrient support, and environmental modifications. Individuals can significantly lower their toxic burden by synergistically reducing exposure (e.g., contaminated water, industrial foods, UPFs) while enhancing hepatic conjugation and renal excretion (via glutathione, minerals, and phytonutrients). Those with occupational exposures or genetic detoxification impairments should pursue professional consultation to balance metal mobilisation with excretion capacity, preventing redistribution to sensitive tissues like the CNS.
Ready to take control of your heavy metal detox journey?
Book a consultation to develop a science-backed plan tailored to your unique exposure risks, genetic profile, and health objectives.