Immunobiotics: The New Frontier in Targeted Gut Health for Immune Resilience
Discover Immunobiotics: the next-generation probiotics scientifically selected to directly modulate your immune system. Learn how they work, their proven benefits, and how they differ from synbiotics, psychobiotics, and postbiotics.
For decades, the conversation around probiotics has focused on digestion. We’ve been told these "good bacteria" are essential for a happy gut, aiding everything from bloating and regularity to brain health, mood, and resilience. While this is true, a profound scientific evolution is redefining our understanding. Emerging from cutting-edge microbiome research is a new, more sophisticated category: Immunobiotics.
This isn't just a marketing buzzword; it's a paradigm shift. Immunobiotics are a specific class of probiotic strains that have been scientifically selected and validated for a primary function: directly and beneficially modulating the human immune system. Moving beyond general gut support, they offer a targeted approach to improving immune resilience, managing inflammation, and maintaining the integrity of our most critical barrier—the gut lining.
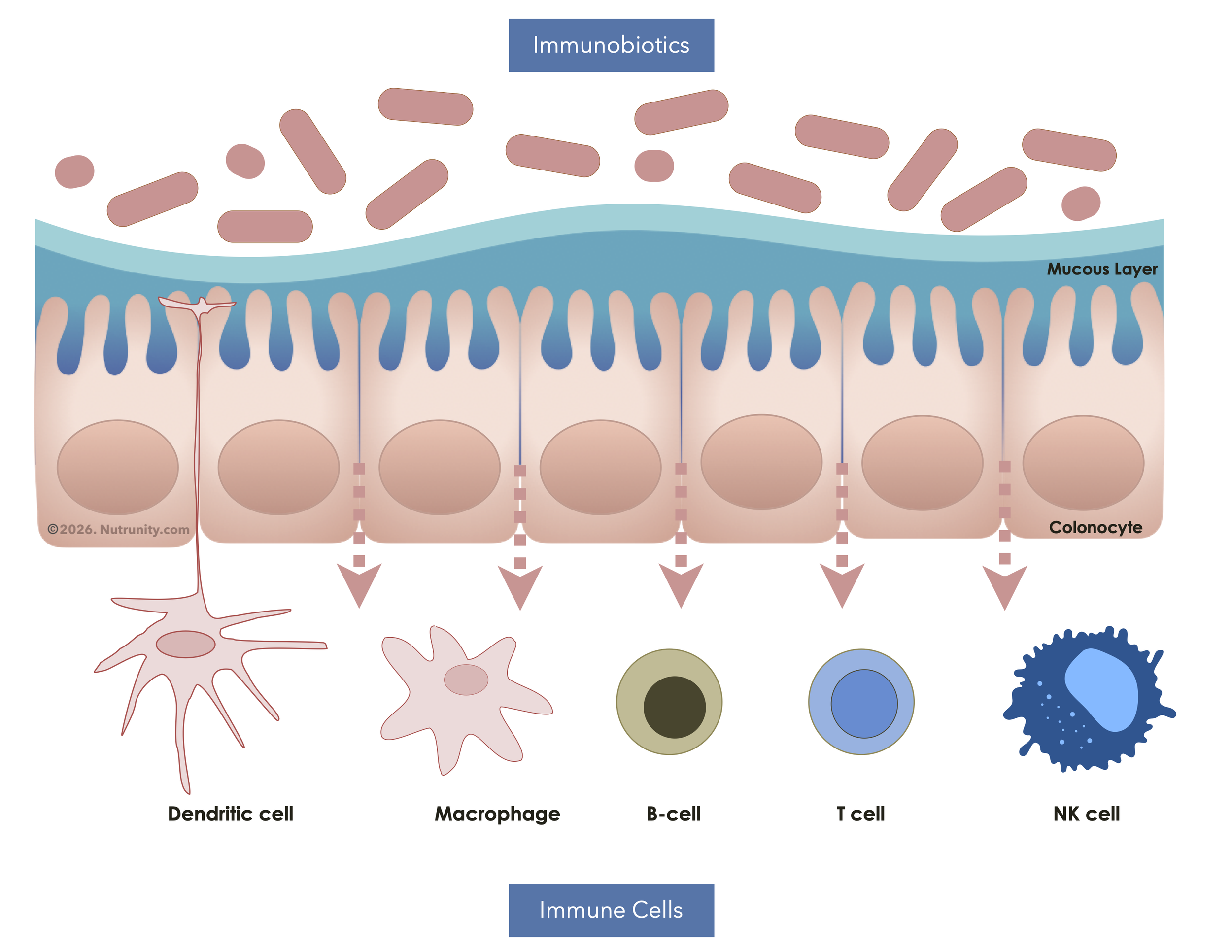
Effect of Immunobiotics on specific immune cells
Yet another “‑biotic” term: What are immunobiotics?
In recent years, the vocabulary of gut‑microbe science has exploded: probiotics, prebiotics, postbiotics, psychobiotics, symbiotics, paraprobiotics and, more recently, immunobiotics. Amid this terminology boom, immunobiotics are not just a marketing rebrand but a more precise way of thinking about how certain microbes interact with the immune system.
The term “immunobiotic” was proposed to describe specific microbial strains that beneficially regulate host immunity, particularly at mucosal surfaces such as the gut. Unlike generic “probiotics”, which may act mainly through local effects such as competitive exclusion or short‑chain fatty acid production, immunobiotics are selected for their capacity to modulate immune‑cell signalling, cytokine profiles and mucosal barrier function in a targeted, strain‑specific manner.
“Immunobiotics are a specific class of probiotic strains that have been scientifically selected and validated for a primary function: directly and beneficially modulating the human immune system.”
The Gut-Immune Axis: Where 70% of Your Immune System Resides
To appreciate immunobiotics, we must first understand the gut-immune axis. The intestinal tract is far more than a digestive tube; it is the body's largest interface with the external environment and houses approximately 70-80% of the body's immune cells.[1] This vast network, known as the gut-associated lymphoid tissue (GALT), is in constant conversation with the trillions of microbes that constitute the gut microbiota.
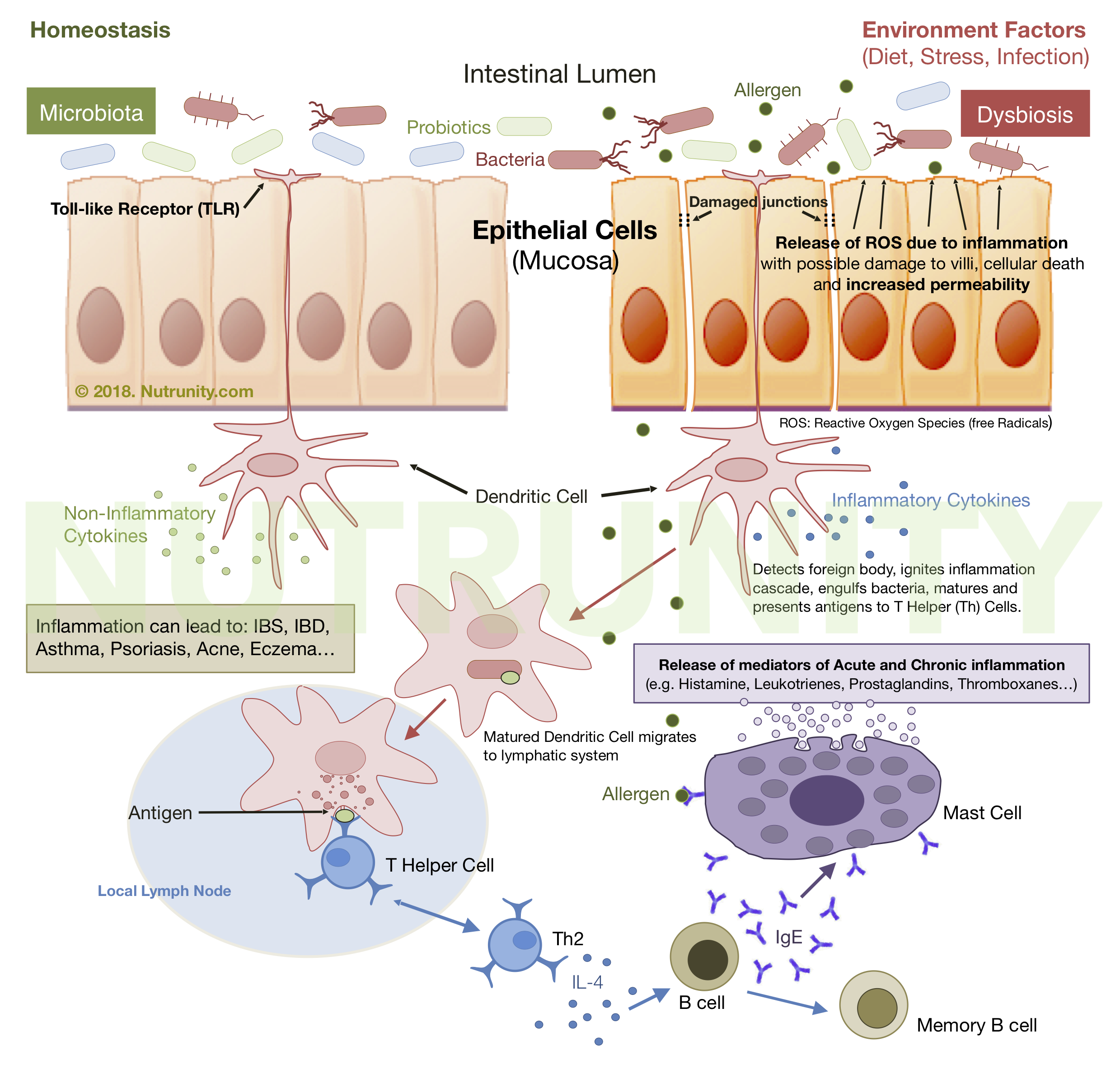
The intestinal epithelium (the lining of the gut) is a dynamic, selective barrier patrolled by immune sentinels (sIgAs). Its role is to efficiently absorb nutrients, to form a robust physical and chemical barrier against pathogens and toxins, and to "educate" the immune system, teaching it to tolerate harmless substances (like food and commensal bacteria) while mounting precise attacks against genuine threats.[2] When this delicate balance is disrupted (a state known as dysbiosis), the consequences can include systemic inflammation, autoimmune reactivity, and increased susceptibility to infection.[3]
What Are Immunobiotics? A Scientific Definition
Immunobiotics are defined as live microbial strains that, when administered in adequate amounts, directly interact with cells of the gut-associated immune system to regulate their function, promoting homeostasis and enhancing defensive responses against pathogens.[4]
The key differentiator from conventional probiotics lies in the specificity of the interaction and the measurable immune outcome. While many probiotics may confer immune benefits as a secondary effect, immunobiotics are characterised by a well-documented, strain-specific ability to:
Interact with Pattern-Recognition Receptors (PRRs):
Immune and epithelial cells are equipped with PRRs, including Toll-like receptors (TLRs) and NOD-like receptors. Immunobiotic strains possess specific microbial-associated molecular patterns (MAMPs) that bind to these receptors, initiating a controlled signalling cascade (5).
Modulate Intracellular Signalling Pathways:
This interaction primarily affects key pathways, including NF-κB and MAPK, which are master regulators of inflammation. Immunobiotics are shown to fine-tune these pathways, preventing their excessive activation, which is linked to chronic inflammatory diseases.[6]
Shape Cytokine Profiles and Immune Cell Responses:
By modulating signalling, immunobiotics direct the immune system’s communication. A hallmark is their ability to promote anti-inflammatory cytokines, such as IL-10, while appropriately regulating pro-inflammatory cytokines, including TNF-α, IL-12, and IL-6.[7] They also influence dendritic cells (the immune system’s “generals”) to promote a balanced T-cell response, favouring regulatory T-cells (Tregs) for tolerance over excessive Th1 or Th17 activation linked to autoimmunity.[8].
Enhance the Mucosal Barrier:
At the interface, specific immunobiotic strains strengthen the gut barrier by upregulating tight junction protein production (e.g., occludin, zonulin-1) and stimulating secretory IgA (sIgA) secretion, the first line of antibody defence in the gut lumen.[9]
Competitively Exclude Pathogens:
By occupying adhesion sites and consuming available nutrients, they help prevent colonisation by pathogenic bacteria.[10]
The Net Clinical Effect: A shift from a state of uncontrolled, low-grade inflammation towards a more efficient, regulated, and responsive immune system. This translates to potential benefits for respiratory infections, gut barrier integrity, inflammatory conditions, and vaccine response.[11]
Dysbiosis and its role in allergies, skin and gut symptoms via immune responses
How immunobiotics talk to the immune system
At the mechanistic level, immunobiotics interact with pattern‑recognition receptors (PRRs) on epithelial and immune cells, including Toll‑like receptors (TLRs) and other sensors that detect microbial‑associated molecular patterns (MAMPs). This interaction triggers intracellular signalling cascades, such as the NF‑κB and MAPK pathways, which, in turn, regulate the expression of cytokines, chemokines, and adhesion molecules.
Depending on the strain and context, immunobiotics can either dampen excessive inflammation (for example, by increasing anti‑inflammatory IL‑10 and reducing TNFα) or increase protective responses (such as IL‑12 and type‑I interferons), thereby steering the immune system away from uncontrolled activation and towards a more balanced, regulated state. In animal and human studies, selected immunobiotic strains have been shown to attenuate intestinal inflammation, reduce the severity of viral infections and improve outcomes in models of colitis and mucosal injury.
A key feature of immunobiotics is their action at the mucosal interface, where they influence both innate and adaptive immunity. At the gut epithelium, they can:
Enhance production of secretory IgA (sIgA), the predominant antibody class in the gastrointestinal tract, which contributes to “immune exclusion” of pathogens and helps maintain homeostasis with commensal microbiota.
Strengthen tight junctions and other components of the epithelial barrier to reduce paracellular permeability and limit bacterial translocation.
Compete with pathogens for adhesion sites and nutrients, effectively excluding harmful microbes from colonising the mucosa.
Key Immunobiotic Strains and Their Evidence-Based Roles
The effects of immunobiotics are highly strain-specific. Not all Lactobacillus or Bifidobacterium strains function as immunobiotics. Below are some of the most clinically studied genera and specific strains.
Lactobacillus rhamnosus GG (LGG): One of the most extensively researched strains. LGG has been shown to enhance mucosal sIgA responses, reduce the incidence and duration of respiratory tract infections in children and adults, and support gut barrier function.[12, 13]
Lactobacillus casei Shirota (LcS): Known for its ability to modulate NK cell activity and improve immune markers in the elderly, a population often experiencing immunosenescence.[14]
Bifidobacterium animalis subsp. lactis BB-12: Clinical trials demonstrate its capacity to support immune function, including increased antibody production following vaccination (e.g., influenza vaccine).[15]
Lactobacillus plantarum 299v (LP299v): This strain has a strong affinity for the intestinal mucosa and has been shown to reduce markers of systemic inflammation (like CRP) and improve gut barrier function in conditions like irritable bowel syndrome (IBS).[16]
Saccharomyces cerevisiae var. boulardii CNCM I-745: A beneficial yeast with potent immunomodulatory effects, particularly in managing antibiotic-associated and C. difficile diarrhoea, partly through modulation of host immune pathways and secretory IgA.[17]
Immunobiotics versus probiotics
The concept of immunobiotics arose partly because the broad term “probiotic” does not distinguish strains that act mainly through local metabolic effects from those that exert strong, specific immune‑modulatory actions.
Immunobiotics are, in essence, a subset of probiotics that have been intentionally selected and characterised for their immuno-modulatory properties, often using in vitro immune‑cell assays and animal models of inflammation or infection. This conceptual refinement allows researchers and clinicians to think more precisely about which strains may be appropriate for conditions driven by immune dysregulation, such as inflammatory bowel disease, allergic disorders, recurrent infections or mucosal injury, rather than using probiotics as a one‑size‑fits‑all category.
Differentiating the “Biotics” from Symbiotics, Psychobiotics, Postbiotics, and More
The field of microbiome science has spawned a new vocabulary. Here’s a clear definition of key terms:
Probiotics: Live microorganisms that, when administered in adequate amounts, confer a health benefit on the host. This is the broad umbrella category.
Prebiotics: A substrate that is selectively utilised by host microorganisms, conferring a health benefit. These are typically non-digestible fibres (e.g., inulin, FOS, GOS) that act as food for beneficial bacteria.[18]
Symbiotics: A synergistic combination of probiotics and prebiotics. The premise is that the prebiotic component selectively supports the survival and implantation of the co-administered probiotic strain(s).[19]
Postbiotics: Preparation of inanimate microorganisms and/or their components that confers a health benefit on the host.[20] These are non-viable bacterial products or metabolic byproducts (e.g., short-chain fatty acids, such as butyrate, or bacterial fragments) that exhibit biological activity. They offer a stable, non-viable alternative to live probiotics.
Psychobiotics: A subcategory of probiotics (and to some extent, prebiotics) that, when ingested in adequate amounts, produce a beneficial effect on mental health (e.g., anxiety, depression, mood) by interacting with the gut-brain axis via neural, endocrine, and immune pathways.[21] Example: Strains like Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 have been shown to reduce psychological distress.
Paraprobiotics (also called non‑viable or inactivated probiotics): often described as non‑viable microbial cells or crude extracts that exert beneficial effects, particularly on immune and barrier function.
Key Differentiation: While a psychobiotic primarily acts through the gut-brain axis to influence mood, an immunobiotic acts through the gut-immune axis to influence systemic and mucosal immunity. A strain could theoretically have properties of both, but research typically categorises them by their primary, most potent mechanism of action.
Some postbiotic and paraprobiotic preparations show immunomodulatory activity similar to that of live immunobiotics, including modulation of NF‑κB/MAPK signalling, cytokine profiles and barrier integrity, but without the need for viable organisms. This opens therapeutic possibilities for individuals who cannot tolerate live probiotics or for whom stability and safety of inactivated products are advantageous.
Why the distinction matters for clinical practice
For practitioners working in functional medicine, nutrition and gastroenterology, understanding these terms is not merely semantic; it has practical implications for strain selection, dosing and outcome expectations.
Immunobiotics are particularly relevant when the clinical picture involves mucosal inflammation, recurrent infections, or immune‑mediated gut disorders, and when the goal is to rebalance cytokine profiles and barrier function in a strain‑specific way.
Symbiotics may be preferred when the aim is to support general microbiota composition and fermentation, especially in individuals with low fibre intake or dysbiosis.
Psychobiotics are appropriate when mood, anxiety or stress‑related symptoms are prominent and linked to gut–brain‑axis dysfunction.
Postbiotics and paraprobiotics offer options for patients who are immunocompromised, critically ill, or otherwise at risk from live‑microbe products, while still aiming to influence immune and barrier function.
Clinical Applications and Considerations for Immunobiotic Use
The potential applications for immunobiotics are vast and grounded in a growing body of clinical research:
Supporting Respiratory Health: Multiple meta-analyses support the use of specific immunobiotic strains (like LGG and Bifidobacterium lactis) in reducing the risk and duration of common upper respiratory tract infections, particularly in children, the elderly, and stressed individuals.[22]
Managing Inflammatory Gut Conditions: In conditions such as IBS and ulcerative colitis, certain immunobiotics can help downregulate gut inflammation, reduce symptom scores, and support barrier repair.[23]
Enhancing Vaccine Efficacy: Co-administration with vaccines (e.g., influenza, rotavirus) has been shown in some studies to improve antibody titres and seroconversion rates, suggesting an adjuvant-like effect.[24]
Combating Immunosenescence: In ageing populations, selected strains can help counteract the natural decline in immune vigilance, improving NK cell activity and response to pathogens.[25]
Important Considerations:
Strain-Specificity: Effects cannot be generalised. The benefits of Lactobacillus rhamnosus GG are unique to that strain.
Dosage (CFU): Adequate colony-forming units (CFUs), typically in the billions, are required for a clinical effect.
Viability: They must be alive and able to colonise, at least transiently. Quality of product formulation and storage is critical.
Individual Variation: Host factors like baseline microbiome, genetics, diet, and health status influence outcomes.
The Future of Immunobiotics: Personalised Microbiome Medicine
The future lies in personalisation. As research progresses, we are moving towards identifying which specific immunobiotic strain or combination is most effective for an individual's unique microbiome signature and immune phenotype. This could revolutionise the management of allergic, autoimmune, and infectious diseases.
Conclusion
Immunobiotics represent a significant leap forward from the first-generation concept of probiotics for “digestive health.” They are a precise, research-driven tool in the emerging field of immuno-nutrition, offering a targeted strategy to educate and strengthen the immune system at its primary site of function, the gut mucosa. By understanding their specific mechanisms, evidence-based strains, and distinctions from other biotics, healthcare professionals and informed consumers can make more strategic decisions to support immune resilience and systemic health.
References
Wiertsema, SP. van Bergenhenegouwen, J. Garssen, J. et al. (2021). The interplay between the gut microbiome and the immune system in the context of infectious diseases throughout life and the role of nutrition in optimizing treatment strategies. Nutrients. 13(3), 886. doi:10.3390/nu13030886
Vighi, G. Marcucci, F. Sensi, L. et al. (2008). Allergy and the gastrointestinal system. Clinical and Experimental Immunology. 153(Suppl. 1), pp. 3-6. doi:10.1111/j.1365-2249.2008.03713.x
Levy, M. Kolodziejczyk, A. Thaiss, C. et al. (2017). Dysbiosis and the immune system. Nature Reviews Immunology. 17, pp. 219–232. doi:10.1038/nri.2017.7
Villena, J. Kitazawa, H. (2014). Modulation of intestinal TLR4-inflammatory signaling pathways by probiotic microorganisms: Lessons learned from Lactobacillus jensenii TL2937. Frontiers in Immunology. 4, 512. doi:10.3389/fimmu.2013.00512
Lebeer, S. Vanderleyden, J. De Keersmaecker, SC. (2010). Host interactions of probiotic bacterial surface molecules: comparison with commensals and pathogens. Nature Reviews Microbiology, 8(3), pp. 171–184. doi: 10.1038/nrmicro2297
Wang, Y. Liu, Y. Sidhu, A. et al. (2012). Lactobacillus rhamnosus GG culture supernatant ameliorates acute alcohol-induced intestinal permeability and liver injury. American Journal of Physiology. Gastrointestinal and Liver Physiology. 303(1), G32-41. doi:10.1152/ajpgi.00024.2012
Liu, H. Kang, X. Yang, X. et al. (2023). Compound probiotic ameliorates acute alcoholic liver disease in mice by modulating gut microbiota and maintaining intestinal barrier. Probiotics and Antimicrobial Proteins. 15(1), pp. 185-201. doi:10.1007/s12602-022-10005-x
Hardy, H. Harris, J. Lyon, E. et al. (2013). Probiotics, prebiotics and immunomodulation of gut mucosal defences: homeostasis and immunopathology. Nutrients. 5(6), pp. 1869-1912. doi:10.3390/nu5061869
Fong, FLY. Kirjavainen, PV. El-Nezami, H. (2016). Immunomodulation of Lactobacillus rhamnosus GG (LGG)-derived soluble factors on antigen-presenting cells of healthy blood donors. Scientific Reports. 6, 22845. doi:10.1038/srep22845
Karczewski, J. Troost, FJ. Konings, I. et al. (2010). Regulation of human epithelial tight junction proteins by Lactobacillus plantarum in vivo and protective effects on the epithelial barrier. American Journal of Physiology. Gastrointestinal and Liver Physiology. 298(6), G851-G859. doi:10.1152/ajpgi.00327.2009
Corr, SC. Li, Y. Riedel, CU. et al. (2007). Bacteriocin production as a mechanism for the antiinfective activity of Lactobacillus salivarius UCC118. Proceedings of the National Academy of Sciences of the United States of America. 104(18), pp. 7617-7621. doi:10.1073/pnas.0700440104
King, S. Glanville, J. Sanders, ME. et al. (2014). Effectiveness of probiotics on the duration of illness in healthy children and adults who develop common acute respiratory infectious conditions: A systematic review and meta-analysis. British Journal of Nutrition. 112(1), pp. 41-54. doi:10.1017/S0007114514000075
Hojsak, I. Snovak, N. Abdović, S. et al. (2010). Lactobacillus GG in the prevention of gastrointestinal and respiratory tract infections in children who attend day care centers: A randomized, double-blind, placebo-controlled trial. Clinical Nutrition. 29(3), pp. 312-316. doi:10.1016/j.clnu.2009.09.008
Kumpu, M. Kekkonen, RA. Kautiainen, H. et al. (2012). Milk containing probiotic Lactobacillus rhamnosus GG and respiratory illness in children: A randomized, double-blind, placebo-controlled trial. European Journal of Clinical Nutrition. 66(9), pp. 1020-1023. doi:10.1038/ejcn.2012.62
Van Puyenbroeck, K. Hens, N. Coenen, S. et al. (2012). Efficacy of daily intake of Lactobacillus casei Shirota on respiratory symptoms and influenza vaccination immune response: A randomized, double-blind, placebo-controlled trial in healthy elderly nursing home residents. American Journal of Clinical Nutrition. 95(5), pp. 1165-1171. doi:10.3945/ajcn.111.026831
Rizzardini, G. Eskesen, D. Calder, PC. et al. (2012). Evaluation of the immune benefits of two probiotic strains Bifidobacterium animalis ssp. lactis, BB-12® and Lactobacillus paracasei ssp. paracasei, L. casei 431® in an influenza vaccination model: A randomised, double-blind, placebo-controlled study. British Journal of Nutrition. 107(6), pp. 876-84. doi:10.1017/S000711451100420X
Ducrotté, P. Sawant, P. Jayanthi, V. (2012). Clinical trial: Lactobacillus plantarum 299v (DSM 9843) improves symptoms of irritable bowel syndrome. World Journal of Gastroenterology. 18(30), pp. 4012-4018. doi:10.3748/wjg.v18.i30.4012
Kelesidis, T. Pothoulakis, C. (2012). Efficacy and safety of the probiotic Saccharomyces boulardii for the prevention and therapy of gastrointestinal disorders. Therapeutic Advances in Gastroenterology. 5(2), pp. 111-125. doi:10.1177/1756283X11428502
Gibson, GR. Hutkins, R. Sanders, ME. et al. (2017). Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nature Reviews Gastroenterology & Hepatology. 14(8), pp. 491-502. doi:10.1038/nrgastro.2017.75
Swanson, KS. Gibson, GR. Hutkins, R. et al. (2020). The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nature Reviews Gastroenterology & Hepatology. 17(11), pp. 687-701. doi:10.1038/s41575-020-0344-2
Salminen, S. Collado, MC. Endo, A. et al. (2021). The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nature Reviews Gastroenterology & Hepatology. 18(9), pp. 649-667. doi:10.1038/s41575-021-00440-6
Dinan, TG. Stanton, C. Cryan, JF. (2013). Psychobiotics: A novel class of psychotropic. Biological Psychiatry. 74(10), pp. 720-726. doi:10.1016/j.biopsych.2013.05.001
Zhao, Y. Dong, BR. Hao, Q. (2022). Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Systematic Reviews. 8(8), CD006895. doi:10.1002/14651858.CD006895.pub4
Derwa, Y. Gracie, DJ. Hamlin, PJ. et al. (2017). Systematic review with meta-analysis: The efficacy of probiotics in inflammatory bowel disease. Alimentary Pharmacology & Therapeutics. 46(4), pp. 389-400. doi:10.1111/apt.14203
Yeh, TL. Shih, PC. Liu, SJ. et al. (2018). The influence of prebiotic or probiotic supplementation on antibody titers after influenza vaccination: A systematic review and meta-analysis of randomized controlled trials. Drug Design, Development and Therapy. 12, pp. 217-230. doi:10.2147/DDDT.S155110
Akatsu, H. Arakawa, K. Yamamoto, T. et al. (2013). Lactobacillus in jelly enhances the effect of influenza vaccination in elderly individuals. Journal of the American Geriatric Society. 61(10), pp. 1828-1830. doi:10.1111/jgs.12474
Clancy, R. (2003). Immunobiotics and the probiotic evolution. FEMS Immunology & Medical Microbiology. 38(1), pp. 9–12. doi:10.1016/S0928-8244(03)00147-0
Kanmani, P. Kim, H. (2020). Beneficial effect of immunobiotic strains on attenuation of Salmonella induced inflammatory response in human intestinal epithelial cells. PLoS One. 15(3), e0229647. doi:10.1371/journal.pone.0229647
Mantis, NJ. Rol, N. Corthésy, B. (2011). Secretory IgA's complex roles in immunity and mucosal homeostasis in the gut. Mucosal Immunology. 4(6), pp. 603-611. doi:10.1038/mi.2011.41
Oxford Dictionary. Definition of symbiotic. Available at: https://www.oxfordlearnersdictionaries.com/us/definition/english/symbiotic
León, ED. Francino, MP. (2022). Roles of secretory immunoglobulin A in host-microbiota interactions in the gut ecosystem. Frontiers in Microbiology. 13, 880484. doi:10.3389/fmicb.2022.880484
Rodriguez, AV. Griet, M. (2015). NF-κB in anti-Inflammatory activity of probiotics: An update. Current Immunology Reviews. 12(1). Available: https://ri.conicet.gov.ar/bitstream/handle/11336/29078/CONICET_Digital_Nro.25033.pdf?sequence=1&isAllowed=y. [Accesed: 29 Jan. 2026]
Sarkar, A. Lehto, SM. Harty, S. et al. (2016). Psychobiotics and the manipulation of bacteria–gut–brain signals. Trends in Neurosciences. 39(11), pp. 763 - 781. doi:10.1016/j.tins.2016.09.002
Siciliano, RA. Reale, A. Mazzeo, MF. et al. (2021). Paraprobiotics: A new perspective for functional foods and nutraceuticals. Nutrients. 13(4), 1225. doi:10.3390/nu13041225
Vinderola, G. Sanders, ME. Salminen, S. (2022). The concept of postbiotics. Foods. 11(8), 1077. doi:10.3390/foods11081077