The Oestrobolome: How Your Gut Bacteria Control Oestrogen and Why It Matters for Your Brain
When we talk about hormones, we usually think of the ovaries, the adrenal glands, the thyroid, or even HRT or PMS. We rarely think of the colon and, to a greater extent, mitochondria*. But emerging clinical evidence is forcing us to expand our view. Deep within your gut, trillions of bacteria are performing a function that directly influences your hormonal balance, your metabolic health, and even the long-term health of your brain. The key player in this story is called the oestrobolome, the part of your gut microbiome that can process and recycle oestrogens.
Recall:
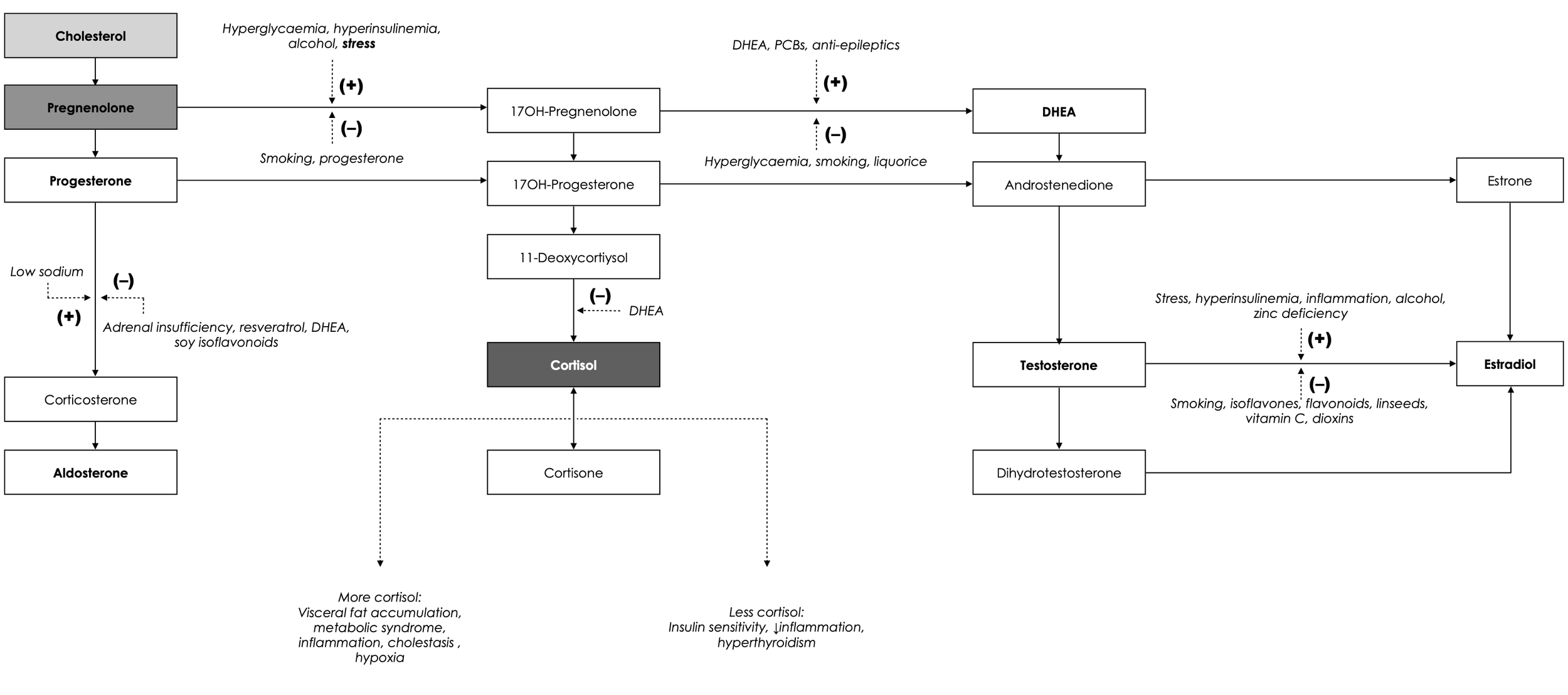
Mitochondria act as the primary site for steroidogenesis, the creation of steroid hormones like oestrogen, progesterone, and testosterone. Mitochondria use the enzyme CYP11A1 to convert cholesterol into pregnenolone, which is the precursor for all sex steroid hormones
Steroidogenesis pathways , with influencing factors. Diagram by Olivier Sanchez, extracted from “Energise - 30 Days to Vitality.” All rights reserved.
The estrobolome is not a single organ. It is a functional subset of your gut microbiome: a specific collection of bacteria that possess the enzyme machinery to metabolise oestrogens. These bacteria, particularly those in genera such as Bifidobacterium and Clostridium, produce an enzyme called beta-glucuronidase. This enzyme can determine whether oestrogen stays in your body or leaves it. Understanding this process is not academic trivia; it is central to understanding a wide range of conditions that I see in clinical practice daily: hormonal imbalances, stubborn weight gain (or weight loss resistance), mood disorders, and even cognitive decline.
Let me walk you through exactly how this works, why it matters for your health, and what you can do about it.
The Oestrogen Lifecycle: From Liver to Gut and Back Again
Estrogen is made mainly in the ovaries (and, to a lesser extent, in fat tissue and the adrenal glands), but to understand the oestrobolome, you first need to understand what happens to oestrogen after it has done its job in your body. Your liver plays a critical role in hormone detoxification. It takes circulating oestrogens (i.e., oestradiol, oestrone, and oestriol) and marks them for elimination through a process called conjugation. Essentially, the liver attaches a glucuronic acid molecule to the oestrogen, creating a water-soluble compound that can be excreted into bile and eliminated from the body.
This is where the story should end. The “used” estrogen should leave your body in your stool.
But here is where your gut bacteria come into play. The beta-glucuronidase enzymes produced by your oestrobolome bacteria can perform the opposite reaction. They can deconjugate that oestrogen, snipping off the glucuronic acid tag. When this happens, the oestrogen is no longer water-soluble. It becomes fat-soluble again, is reabsorbed through the intestinal wall, and re-enters your bloodstream.
Think about the implications of this. Your gut bacteria have the power to override your liver’s decision to eliminate oestrogen. They can effectively recycle hormones back into circulation. This is not inherently bad (some estrogen recycling is normal), but when the system becomes unbalanced, problems arise. This is chronic constipation and a toxic colon take centre stage.
Dysbiosis and Oestrogen Dominance
When the oestrobolome is healthy and diverse, beta-glucuronidase activity is balanced, and oestrogen excretion proceeds normally. But when dysbiosis sets in (the imbalance in the gut microbial community), the picture changes dramatically. An overgrowth of bacteria that produce high levels of beta-glucuronidase leads to excessive oestrogen deconjugation and reabsorption.
The clinical result is a condition often referred to as “oestrogen dominance.” This is not a diagnosis you will find in every medical textbook, but it is a physiological reality that explains a constellation of symptoms: heavy or painful periods, breast tenderness, PMS, fibroids, endometriosis, and even an increased risk of hormone-sensitive cancers like breast and endometrial cancer. The excess oestrogen circulating in the body continues to stimulate tissues long after it should have been eliminated.
Oestrogen-dominance syndrome is being accepted more and more in the medical realm and will help in the prevention of many cancers.
But oestrogen dominance is not just a reproductive issue. It has systemic consequences. Elevated oestrogen levels have been linked to weight gain, particularly around the hips and thighs, and to metabolic disturbances. The hormone is intimately involved in glucose homeostasis and insulin sensitivity. When oestrogen signalling goes awry, metabolism follows suit.
The SCFA Connection: Feeding the Gut That Feeds the Hormones
The oestrobolome does not operate in isolation. Its health and activity are profoundly influenced by the availability of short-chain fatty acids (SCFAs), which are produced when gut bacteria ferment dietary fibre.
SCFAs (i.e., butyrate, acetate, and propionate) are the primary fuel source for the cells lining your colon. They strengthen the gut barrier, reduce inflammation, and create an environment where beneficial bacteria can thrive. A healthy, SCFA-rich environment supports a balanced oestrobolome with appropriate beta-glucuronidase activity. Conversely, a low-fibre diet starves the bacteria that produce SCFAs, leading to a compromised gut lining and a shift toward a dysbiotic, pro-inflammatory microbial community.
This is where the concept of the gut-hormone axis becomes clinically actionable. By eating fibre, you feed the bacteria that produce SCFAs. Those SCFAs nourish the gut lining and support a healthy oestrobolome. A healthy eostrobolome ensures that oestrogens are metabolised appropriately and excreted efficiently. The entire system is interconnected.
Gut Motility: The Unsung Hero of Hormone Clearance
There is another mechanical factor at play here, one that connects back to our earlier discussion of serotonin. Gut motility (the rhythmic muscular contractions that move contents through your digestive tract) is critical for hormone balance.
Think about it logically. The liver packages oestrogen for excretion and sends it to the gut via bile. If that bile and its contents linger in the colon for an extended period (e.g., you are constipated or have slow transit time), the oestrobolome bacteria have more opportunity to deconjugate and recycle those oestrogens. The longer the waste sits, the more reabsorption occurs.
This is why regular bowel movements are not just about comfort. They are a primary pathway for hormone detoxification. I tell my clients that a bowel movement once daily is not optional; it is a hormonal necessity. And as we discussed in the previous article, regular bowel movements depend on adequate mechanical stimulation from dietary fibre and the serotonin release triggered by that stimulation. It all circles back.
Gut-Derived Oestrogen and the Brain: Neuroinflammation and Neurodegeneration
Perhaps the most fascinating and clinically urgent frontier in this research is the connection between gut-derived oestrogen signalling and the brain. Oestrogen is not just a reproductive hormone. It is a potent neurosteroid with profound effects on the central nervous system.
Oestrogen receptors (particularly ER-beta and GPER1) are widely distributed throughout the brain. When activated appropriately, they exert neuroprotective effects. They support synaptic plasticity, promote the expression of brain-derived neurotrophic factor (BDNF), and help regulate the stress response via the hypothalamic-pituitary-adrenal (HPA) axis. Oestrogen also has potent anti-inflammatory effects in the brain. Research has demonstrated that ER-beta signalling can negatively regulate microglia-mediated inflammation, essentially calming the brain’s immune cells.
But here is the problem. When the oestrobolome is dysbiotic, and oestrogen metabolism is disrupted, the signalling to the brain becomes chaotic. The ratio of different oestrogen metabolites changes. The balance between ER-alpha and ER-beta activation shifts, often toward ER-alpha dominance, which can be pro-inflammatory rather than protective.
This disruption has been linked to several neuropsychiatric and neurodegenerative conditions. Depression, anxiety, and postpartum mood disorders all show connections to dysregulated oestrogen signalling and gut microbiome composition. The dramatic hormonal shifts that occur during perimenopause and menopause, combined with age-related changes in the gut microbiome, may create a perfect storm for neuroinflammation.
The implications for neurodegeneration are equally concerning. Parkinson’s disease, for example, has well-documented connections to both gut dysfunction and oestrogen signalling. The pathology of Parkinson’s may actually begin in the gut decades before motor symptoms appear. Disrupted gut microbiota, including an imbalanced oestrobolome, could contribute to an inflammatory environment that sets the stage for neurodegeneration. The link to heavy metals is also well-documented, particularly for lead and mercury, including their impact on the gut environment, their accumulation in the brain, and their interaction with aluminium, causing neurodegenerative disorders, including Alzheimer’s disease.
Alzheimer's disease also shows striking sex differences and connections to oestrogen. Women are disproportionately affected by Alzheimer's, and the dramatic drop in oestrogen during menopause is implicated in this increased risk. But it may not be just ovarian oestrogen. The gut's contribution to overall oestrogen status and inflammation (and mitochondrial health) could be a modifiable factor in neuroprotection.
“Oestrogen receptors are widely distributed throughout the brain. When activated appropriately, they exert neuroprotective effects. They support synaptic plasticity, promote the expression of brain-derived neurotrophic factor (BDNF), and help regulate the stress response via the hypothalamic-pituitary-adrenal (HPA) axis. Oestrogen also has potent anti-inflammatory effects in the brain. Research has demonstrated that ER-beta signalling can negatively regulate microglia-mediated inflammation, essentially calming the brain’s immune cells.”
How Diet Shapes the Oestrobolome
So, what determines whether your oestrobolome is working for you or against you?
The answer is: Diet.
Fibre is the non-negotiable foundation. When you eat a variety of fibres from whole plant foods (e.g., vegetables, fruits, legumes, nuts, seeds, and whole grains), you feed the diverse community of bacteria that produce SCFAs and help maintain a healthy gut environment. This includes the bacteria of the oestrobolome.
Specific fibres may have particular benefits. Flaxseeds, for example, contain lignans that bacteria convert into enterolignans, which can weakly bind to oestrogen receptors and modulate oestrogen activity. Cruciferous vegetables like broccoli contain compounds that support liver detoxification pathways, helping the liver process oestrogen more efficiently before it even reaches the gut. For a more potent functional food, think sprouted organic purple broccoli or kale.
The standard Western diet, low in fibre and high in ultra-processed foods, fat, and sugar, produces the opposite effect. It promotes the growth of pro-inflammatory bacteria, reduces SCFA production, weakens the gut barrier, and disrupts the oestrobolome. The result is increased intestinal permeability, systemic inflammation, and dysregulated oestrogen recycling, all of which contribute to the conditions described above.
Clinical Takeaways: What This Means for You
As a naturopath, my approach to hormonal health always begins in the gut. Before reaching for herbs or hormones, I look at the foundation. Here is what I want you to take away from this discussion.
First, your gut bacteria are active participants in your hormonal health. The oestrobolome is a real and powerful mediator of oestrogen metabolism. If you are struggling with symptoms of oestrogen dominance (e.g., PMS, heavy periods, fibroids, endometriosis), your gut health deserves scrutiny.
Second, fibre is your best friend. The recommendation of 25 to 35 grams of fibre per day is not arbitrary. It is the amount required to feed a healthy microbiome, support regular bowel movements, and ensure efficient hormone excretion. If you are not meeting this target, your hormonal detoxification is likely compromised.
Third, variety matters. Different fibres feed different bacteria. Eating the same few vegetables every day will not cultivate the diverse microbiome that supports a healthy oestrobolome. Aim for a rainbow of plant foods daily and weekly, different colours, different types, different fibres. For example, on day one, you can have asparagus, linseeds, and buckwheat; on day two, resistant starch like cooked-and-cooled potatoes, tiger nuts, and plantains; and on day three, a chia seed pudding for breakfast, and for lunch, a quinoa salad with plenty of different vegetables, and for dinner, a large portion of broccoli or kale with your meal.
Fourth, motility is critical. A sluggish colon gives bacteria more time to recycle oestrogen. Support regular bowel movements with adequate fibre, hydration, and movement. If you are chronically constipated, you are likely reabsorbing hormones that should be leaving your body.
Finally, the gut-brain connection runs through hormones. The link between gut health, oestrogen metabolism, and brain health is one of the most exciting frontiers in medicine. Protecting your brain as you age may depend as much on what happens in your colon as on your intake of omega-3s. By supporting your oestrobolome with a fibre-rich diet, you are not just balancing your hormones, you are potentially safeguarding your cognitive future.
References
Atoum, M., Padma, K. (2025). Gut microbiota-estrogen axis: Its influence on female health outcomes – A narrative review. Acta Biomedica. 96(1), 15980. doi:10.23750/abm.v96i1.15980
Caldarelli, M., Rio, P., Marrone, A. et al. (2024). Gut-brain axis: Focus on sex differences in neuroinflammation. International Journal of Molecular Science. 25(10), 5377. doi:10.3390/ijms25105377
Church, JS., Bannish, JAM., Adrian, LA. et al. (2023). Serum short chain fatty acids mediate hippocampal BDNF and correlate with decreasing neuroinflammation following high pectin fiber diet in mice. Frontiers in Neuroscience. 17, 1134080. doi:10.3389/fnins.2023.1134080
Ervin, SM., Li, H., Lim, L. et al. (2019). Gut microbial β-glucuronidases reactivate estrogens as components of the estrobolome that reactivate estrogens. Journal of Biological Chemistry. 294(49), pp. 18586-18599. doi:10.1074/jbc.RA119.010950
Hu, S., Ding, Q., Zhang, W. et al. (2023). Gut microbial beta-glucuronidase: A vital regulator in female estrogen metabolism. Gut Microbes. 15(1), 2236749. doi:10.1080/19490976.2023.2236749
La Torre, D., Verbeke, K., Dalile, B. (2021). Dietary fibre and the gut-brain axis: microbiota-dependent and independent mechanisms of action. Gut Microbiome (Camb). 2, e3. doi:10.1017/gmb.2021.3
Larnder, AH., Manges, AR., Murphy, RA. (2025). The estrobolome: Estrogen-metabolizing pathways of the gut microbiome and their relation to breast cancer. International Journal of Cancer. 157(4), pp. 599-613. doi:10.1002/ijc.35427
Lu, J., Xian, TJ., Li, CJ. et al. (2025). The estrogen-brain interface in neuroinflammation: A multidimensional mechanistic insight. Frontiers in Aging Neuroscience. 17, 1671552. doi:10.3389/fnagi.2025.1671552
Mora-Ortiz, M., García-Fernández, H., Perez-Martinez, P. et al. (2024). The gut microbiota-brain axis role in neurodegenerative diseases and implications according to the sex. Open Research Europe. 4, 230 doi:10.12688/openreseurope.18568.1
Saha, P., Weigle, IQ., Slimmon, N. et al. (2024). Early modulation of the gut microbiome by female sex hormones alters amyloid pathology and microglial function. Scientific Reports. 14, 1827. doi:10.1038/s41598-024-52246-6
Villa, A., Vegeto, E., Poletti, A. et al. (2016). Estrogens, neuroinflammation, and neurodegeneration. Endocrine Reviews. 37(4), pp. 372-402. doi: 10.1210/er.2016-1007