Mitochondrial Health: How Stress Drains Your Cellular Energy and What You Can Do About It
Nowadays, stress often feels like the price of success. We’re always on the go, pushing through deadlines, juggling responsibilities, and trying to find balance, all the while being glued to screens. Short-term stress can help you focus and thus increase your performance, but long-term (chronic) stress slowly erodes your body’s vitality.
At the centre of the inevitable breakdown? Your mitochondria! These microscopic power plants nestled in nearly every cell of your body are responsible for converting oxygen and nutrients into usable energy (ATP). Though, they have many more functions.
But here’s the catch: chronic stress doesn’t just wear you out mentally—it burns you out biologically, right down to the cellular level.
In this article, we explore the cellular cost of chronic stress, its impact on your mitochondria, and how restoring mitochondrial health can help you regain your energy, focus, and resilience.
It is essential to remember that mitochondria fuel your every thought, movement, and bodily function. However, when stress becomes a persistent companion, it silently “chokes” those tiny organelles, gradually reducing the energy your cells can produce. Understanding how chronic stress disrupts mitochondrial function reveals why fatigue, cognitive fog, and diminished resilience often accompany prolonged pressure, and how restoring cellular energy can renew both mind and body.
What Happens in Your Body During Stress?
Stress, in biological terms, is a state of heightened alert designed to protect you from threats, whether physical danger or psychological pressure.
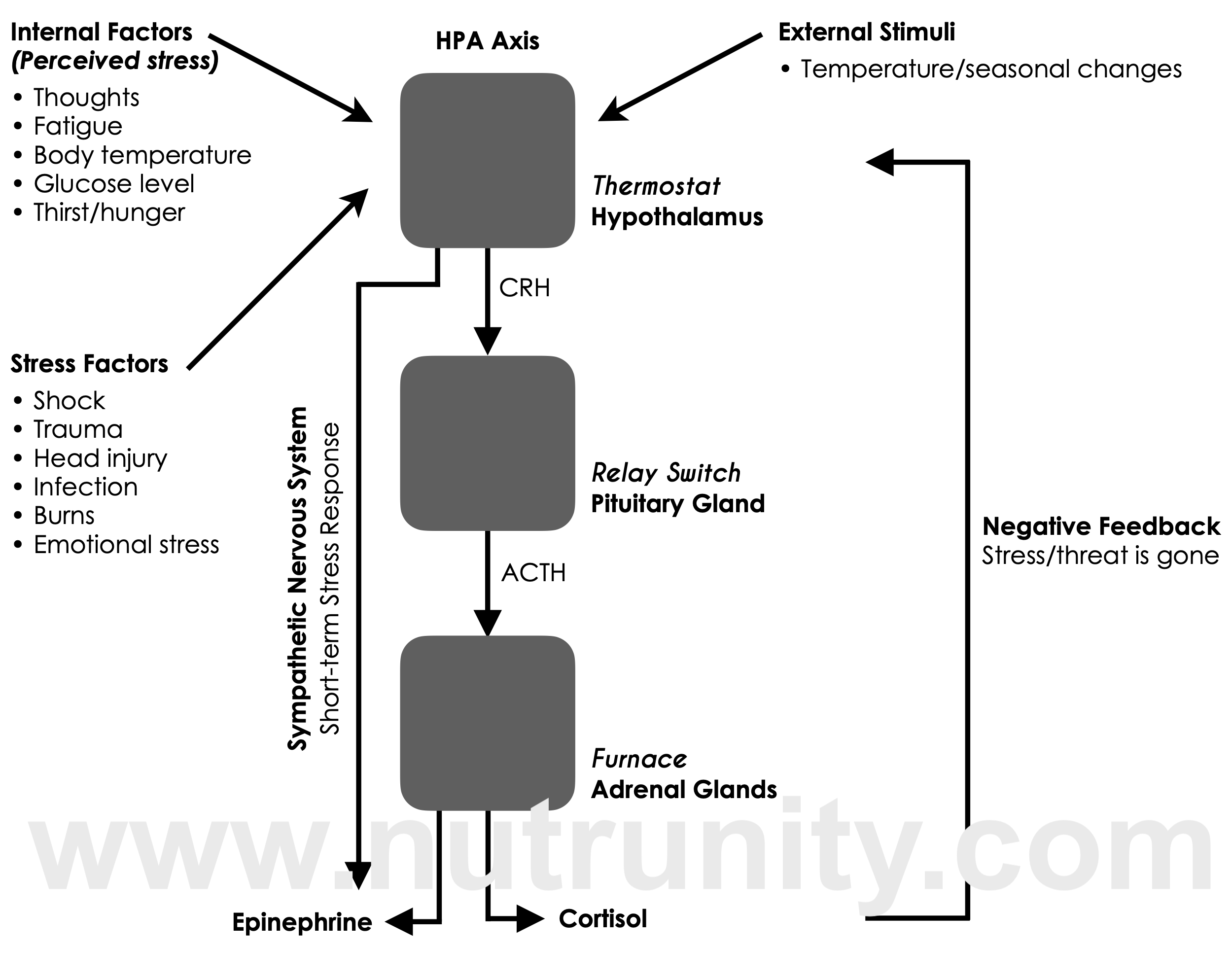
When your brain perceives a stressor (internal or external), it signals the hypothalamic-pituitary-adrenal (HPA) axis, a network linking your brain and endocrine system. This kicks off a hormonal cascade:
The hypothalamus signals the pituitary gland, which in turn activates the adrenal glands
The adrenal glands release epinephrine (adrenaline), norepinephrine (noradrenaline) and, if the stressor is not removed, cortisol.
Epinephrine and norepinephrine spike your heart rate, sharpen your senses (your pupils dilate, so you can see better, the danger or the quickest route to escape), increase blood flow to muscles (to supply energy and nutrients so you can run as fast as possible), and heighten alertness. Cortisol, the primary stress hormone, helps mobilise glucose for quick energy and suppresses systems that aren’t essential in an emergency. This includes digestion, reproduction, and immune function. Survival is the goal!
This system evolved to help early humans respond quickly to acute, immediate danger, like escaping a predator. Once the threat is gone, cortisol levels naturally drop, and the body returns to a state of rest and repair.
However, modern stressors, such as deadlines, financial strain, chronic illness, overexposure to social media, sleep deprivation, or made-up stress (rumination, anxiety, etc.), keep you “switched on.” Cortisol remains elevated, triggering a chain reaction of downstream effects on metabolism, immunity, inflammation (as your digestive capacities are suppressed and food lingers undigested, putrefying and producing gas, while toxins irritate the gut lining, leading to low-grade inflammation and intestinal hyperpermeability, also known as “leaky gut” syndrome), and (critically) your mitochondria.
Why Mitochondria Are Central to Energy, Focus, Mood, and Resilience
Mitochondria are not only power generators; they are also:
In charge of cellular lifespan (via apoptosis, or programmed cell death)
Regulators of inflammation and oxidative stress. Energy production produces a large amount of ROS (reactive oxygen species, commonly known as free radicals), and the body has antioxidant defences to prevent damage to the mitochondria, mitochondrial DNA and our DNA. However, when stress derails our cells’ function, our defences have little in place to avoid mitochondrial, cellular and tissue damage.
Key players in brain health, including neurotransmitter synthesis and synaptic plasticity
Architects of energy-intensive processes like muscle contraction, digestion, and immunity.
These organelles convert macronutrients (from food) and oxygen (from breathing) into ATP (adenosine triphosphate)—the molecule that powers nearly every cellular activity.
A Study in Review
“Psychological stress and mitochondria: A systematic review.”
The systematic review by Picard and McEwen (2018) examines how psychological stress impacts the structure and function of mitochondria, serving as a key interface between psychosocial experiences and biological stress responses. Through a comprehensive review of 23 experimentally controlled studies, the authors demonstrate that both acute and chronic psychological stress have a significant influence on mitochondrial biology, particularly within the brain. Most studies (19 out of 23) reported stress-induced impairments in mitochondrial function, such as altered energy production and increased mitochondrial damage, while a few documented temporary increases in mitochondrial size or activity post-stress.
The review also highlights the concept of mitochondrial allostatic load (MAL), which refers to the maladaptive molecular and functional changes that mitochondria undergo under chronic stress, contributing to widespread biological effects, including inflammation and cellular ageing. Although human research is currently limited to observational studies, which mostly measure indirect mitochondrial markers (such as mitochondrial DNA), the accumulated animal evidence strongly supports the notion that chronic stress leads to significant mitochondrial recalibrations.
Picard and McEwen discuss how mitochondria may sense, integrate, and transduce psychological stress signals into cellular changes affecting energy metabolism, neuroendocrine pathways, and immune function, potentially linking emotional states and psychosocial stressors to physical health outcomes.
In essence, this review demonstrates that chronic psychological stress is not only perceived at the brain level but also induces cellular “wear and tear,” reflected in mitochondrial dysfunction and the physiological consequences of prolonged stress exposure. It establishes mitochondria as a pivotal subcellular hub connecting mental health and systemic biological resilience or vulnerability, offering a promising target for therapeutic interventions aimed at restoring cellular energy balance under stress.
___
Picard, M. McEwen, BS. (2018). Psychological stress and mitochondria: A systematic review. Psychosomatic Medicine. 80(2), pp. 141-153. doi:10.1097/PSY.0000000000000545. PMID: 29389736; PMCID: PMC5901654.
Your brain, despite weighing only about 2% of your body weight, consumes up to 20% of your total energy—making it especially vulnerable to mitochondrial dysfunction. This is why signs like brain fog, memory issues, and emotional dysregulation are early indicators of mitochondrial stress.
“Mitochondria are responsible for providing our cells with energy, as well as regulating oxidative stress and apoptosis, and considerable evidence demonstrates that mitochondria-related alterations are prevalent during chronic stress and depression.”
A Study in Review
“An Energetic View of Stress: Focus on Mitochondria.”
This influential review bridges the fields of stress biology and cellular bioenergetics, proposing a new integrative framework where mitochondria play a central role in how psychological stress influences health and disease over time.
— Mitochondria as Mediators of the Stress Response
Rather than being passive victims of stress, mitochondria detect, interpret, and participate in the stress response. Their function is modulated by:
Hormonal signalling (e.g., glucocorticoids like cortisol)
Neurotransmitters (e.g., norepinephrine)
Inflammatory cytokines
Reactive oxygen species (ROS)
This expands the classical stress response model to include bioenergetic adaptations at the cellular level.
— The Mitochondrial Allostatic Load (MAL) Model
Mitochondrial allostatic load, defined as the gradual wear and tear on mitochondria due to chronic stress exposure (allostatic load), but applied at the cellular level
This includes:
Accumulated mitochondrial DNA damage
Impaired mitochondrial dynamics (fission, fusion, biogenesis)
Decreased ATP output
Dysregulated mitophagy (removal of damaged mitochondria)
Elevated ROS and inflammation
These dysfunctions propagate upstream, ultimately affecting mood, cognition, immunity, and metabolic regulation.
— Bidirectional Loop Between Brain and Mitochondria
The review emphasises the dynamic feedback loop between mitochondria and brain function. The brain regulates mitochondrial activity through neuroendocrine stress signalling, such as activation of the HPA axis. In turn, mitochondrial function influences brain operations via energy availability, neurotransmitter production, redox balance, and inflammatory responses.
Positive or negative feedback loops may support resilience or lead to illness, depending on how flexible and healthy the mitochondria are under stress (and how the stress is perceived).
— Mitochondria and Resilience
The authors propose that mitochondrial fitness is a core biological determinant of stress resilience versus vulnerability. Individuals with healthier mitochondria may:
Adapt more efficiently to psychosocial threats
Recover faster from stress responses
Be protected against neuropsychiatric disorders and metabolic diseases
Clinical and Research Implications
The authors suggest several novel implications:
Targeting mitochondrial function could support mental health and increase stress resilience (via exercise, nutrition, pharmacological agents, or stress management interventions)
Measuring mitochondrial efficiency or damage could be a tool for risk stratification or treatment personalisation
Stress research must integrate bioenergetics and mitochondrial biology to better model real-world health outcomes
___
Picard, M. McEwen, BS. Epel, ES. et al. (2018). An energetic view of stress: Focus on mitochondria. Frontiers in Neuroendocrinology. 49, pp. 72–85. doi:10.1016/j.yfrne.2018.01.001
“By shaping cellular metabolism, mitochondrial function represents an important biological intersection that links stress physiology, neuroplasticity, and systemic health.””
Chronic Stress and the Mitochondrial Burnout Connection
When cortisol remains elevated over time, the effects trickle down through your cells in ways that are invisible, but significant:
1. Disrupted Mitochondrial Biogenesis
Mitochondrial biogenesis is the process by which new mitochondria are formed. An adaptive phenomenon to match the increased energy demands, promote recovery, and enhance resilience. For example, the more you exercise, like running, the more mitochondria you will have in your cells to support endurance.
Chronic stress inhibits this process, reducing your body’s ability to build more energy-generating units and making mitochondria more susceptible to oxidative damage and apoptosis.
2. Increased Oxidative Stress and Mitochondrial Damage
Persistent stress leads to the overproduction of ROS, unstable molecules that attack lipids, proteins, and DNA, including mitochondrial DNA (mtDNA). Damaged mtDNA reduces the ability to produce ATP efficiently, leading to fatigue and other complications.
This creates a vicious cycle. Damaged mitochondria produce more ROS, which damages them further.
3. Impaired Mitophagy: Cellular Housekeeping Breaks Down
Mitophagy is the cellular process of removing dysfunctional mitochondria. When chronic stress interferes with this process, damaged mitochondria accumulate, resulting in reduced energy production and increased inflammatory signals.
This mitochondrial clutter drains energy and raises your risk for neurodegenerative diseases, autoimmunity, and ageing-related decline..
4. Immune System Dysregulation and “Inflammaging”
Mitochondria are intimately connected to the immune system. In a healthy state, they help coordinate immune responses. Under chronic stress, the immune system becomes dysregulated, promoting a state of low-grade, persistent inflammation.
This “inflammaging” state drives mitochondrial dysfunction and is implicated in conditions like depression, cardiovascular disease, and chronic fatigue syndrome (CFS).
— The Usual Symptoms
You may already be experiencing the signs of mitochondrial dysfunction without realising it. Here’s what to watch for:
Never feeling refreshed upon waking up, even after 8+ hours of sleep, and unrelenting fatigue
Brain fog, difficulty concentrating, or memory lapses
Frequent infections or slow recovery from illness
Blood sugar imbalances and increased cravings
Muscle tightness, weakness, or poor endurance
Emotional instability, low mood, or lack of motivation
Poor stress tolerance or heightened reactivity
Trouble falling or staying asleep
These symptoms are often mistaken for just “being busy” or “getting older.” But in many cases, they’re signals that your mitochondria are under pressure.
How does chronic stress affect your body?
When stress persists, cortisol levels stay elevated, keeping your body in a continuous state of alert that disrupts several critical functions. Typically, cortisol levels peak in the morning and decline at night; however, chronic elevation disrupts this balance, leading to sleep problems such as difficulty falling asleep and frequent awakenings. Over time, sustained high cortisol paradoxically fuels inflammation by dysregulating the immune system. Additionally, cortisol alters glucose and fat metabolism, which can contribute to cravings and weight gain. Mentally, prolonged excess cortisol is associated with increased anxiety, irritability, and depression. These interconnected effects place added strain on your mitochondria, the cellular powerhouses responsible for generating the energy your body needs.
HPA axis with short-term stress response, chronic stress, and negative feedback pathway
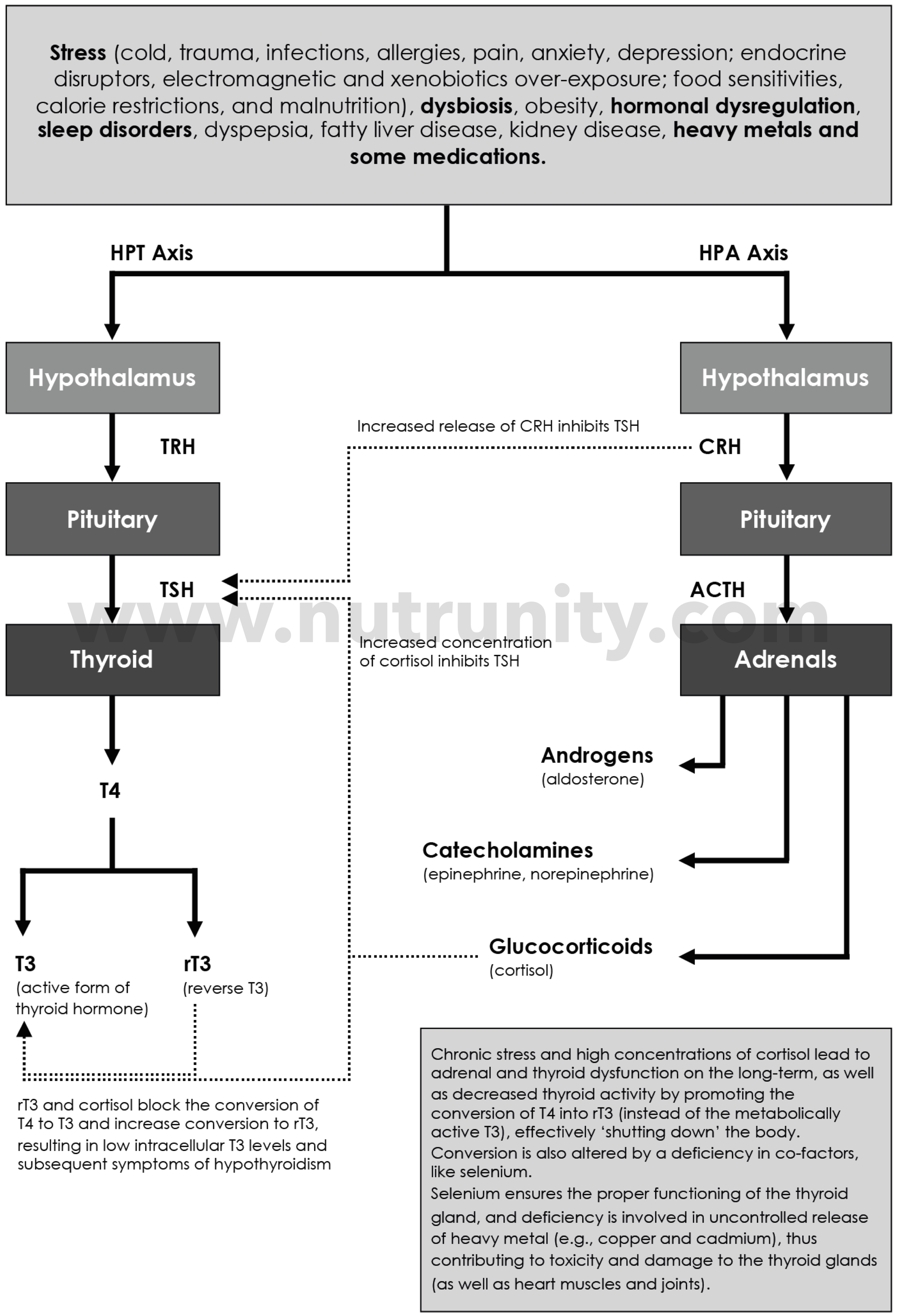
Effect of stress on HPA axis and hormone balance, including rT3 and stress hormones
A Study in Review
“Stress and Psychiatric Disorders: The Role of Mitochondria.”
This review examines the pivotal role mitochondria play in mediating the impact of stress on mental health and psychiatric disorders. The authors present evidence suggesting that mitochondrial dysfunction may be responsible for many of the biological changes seen in depression, anxiety, PTSD, and bipolar disorder, especially those linked to chronic stress.
1. Mitochondria as Stress Sensors and Responders
Mitochondria aren’t just passive energy producers; they sense and respond to psychological and physiological stress
Stress hormones like cortisol and inflammatory cytokines directly influence mitochondrial function, leading to changes in:
Energy production (ATP)
Reactive oxygen species (ROS) generation
Mitochondrial gene expression
2. Early Life Stress & Mitochondrial Programming
Adverse childhood experiences (ACEs) can have lasting effects on mitochondrial structure and function.
These early stressors appear to “programme” mitochondria, increasing susceptibility to future stress and psychiatric illness.
Studies have linked early-life maltreatment to reduced mitochondrial DNA copy number, altered mitochondrial respiration, and increased oxidative stress.
3. Mitochondrial Dysfunction in Psychiatric Illness
Postmortem and peripheral studies show:
Decreased mitochondrial density and impaired bioenergetics in depression and bipolar disorder
Elevated markers of oxidative stress and inflammation
Aberrant mitochondrial DNA (mtDNA) content and function
Psychiatric symptoms such as fatigue, brain fog, emotional dysregulation, and anhedonia (common in depression and PTSD), mirror signs of mitochondrial dysfunction.
4. Psychotropic Medications and Mitochondria
Antidepressants and mood stabilisers influence mitochondrial activity:
Some improve mitochondrial function, increasing ATP availability and reducing oxidative stress.
Others may impair function at high doses or with long-term use.
This interaction may explain variability in treatment response and support the idea of mitochondria as therapeutic targets.
5. Mechanisms Linking Stress, Mitochondria, and Mental Illness
Inflammation, oxidative damage, and metabolic disruption emerge as central pathways. Mitochondria integrate these signalling pathways and determine whether the cell adapts or becomes dysfunctional under stress.
Clinical Implications
Mitochondrial health could serve as a biomarker for stress-related psychiatric disorders and treatment prognosis.
Interventions that enhance mitochondrial function, such as exercise, circadian regulation, antioxidants, and stress management techniques, may improve psychiatric symptoms and resilience.
The review calls for a “mitocentric” model of psychopathology, where mitochondrial dynamics are placed at the heart of stress vulnerability and mental illness.
Conclusion
Daniels et al. make a compelling case that mitochondria are not just affected by psychiatric disorders but might actually drive their development in the context of stress. The bidirectional link between stress, mitochondrial health, and mental health presents new opportunities for diagnostics, preventive strategies, and therapeutic approaches in clinical psychology and psychiatry.
___
Daniels, TE. Olsen, EM. Tyrka, AR. (2020). Stress and psychiatric disorders: The role of mitochondria. Annual Review of Clinical Psychology. 16, pp. 165–186. doi:10.1146/annurev-clinpsy-082719-104030
A Study in Review
“Mitochondria as Key Components of the Stress Response.”
This foundational review outlines an early yet forward-thinking view: mitochondria are integral regulators, not merely effectors, in the body’s response to stress. It proposes that mitochondrial responses directly impact both physiological adaptation and pathophysiological outcomes resulting from acute and chronic stress.
The review establishes a connection between neuroendocrinology (HPA axis), molecular biology, and mitochondrial function, suggesting that mitochondria play a crucial role in determining an organism’s ability to adapt to biological challenges.
1. Mitochondria Are Central to Stress Physiology
Mitochondria provide ATP for stress responses and modulate stress pathways by managing:
Calcium signalling
Steroid hormone biosynthesis (e.g., cortisol production)
Reactive oxygen species (ROS)
Apoptosis (programmed cell death)
These bioenergetic and regulatory functions extend mitochondria’s influence across virtually every organ system affected by stress, including the brain, muscles, and immune system.
2. Bidirectional Feedback Between Stress Hormones and Mitochondria
Glucocorticoids (like cortisol), released during stress via the HPA axis, have direct effects on mitochondria, including:
Altering mitochondrial gene expression
Modifying mitochondrial enzyme activity
Changing organelle shape and number
Mitochondria, in turn, influence glucocorticoid sensitivity, acting as “amplifiers” or “buffers” of the stress response depending on their health and performance.
This feedback loop suggests that mitochondrial dysfunction could result from and contribute to dysregulated stress responses, which are often implicated in depression, anxiety, and chronic fatigue syndrome.
3. Mitochondria and Gene Regulation
Stress activates a cascade of signalling pathways, which affect nuclear and mitochondrial genomes. These changes can alter long-term gene expression patterns at the cellular and systemic levels, impacting:
Inflammation
Immune function
Energy metabolism
Chronic alterations in gene regulation resulting from this stress-mitochondria crosstalk may partially explain why early life adversity has lasting effects on physical and mental health.
4. Adaptation vs. Maladaptation
The mitochondrial response to stress is designed to be adaptive, providing rapid energy, buffering oxidative insults, and facilitating short-term survival. However, chronic activation leads to mitochondrial failure, manifesting as:
Increased oxidative stress
Mitochondrial DNA (mtDNA) damage
Impaired ATP production
Accelerated cell ageing
These cumulative effects leave the body more vulnerable to disease states, including:
Major depressive disorder
Neurodegenerative conditions (e.g., Parkinson’s, Alzheimer’s)
Metabolic diseases (e.g., diabetes)
Chronic inflammation and autoimmune disorders
5. Therapeutic Potential
The authors argue that protecting mitochondrial function could improve resilience to chronic stress and reduce the risk of developing stress-related diseases. Suggested strategies include:
Antioxidants (to reduce oxidative damage)
Nutrients that support mitochondrial metabolism (e.g., coenzyme Q10)
Hormonal therapies (cautiously, due to glucocorticoid interactions)
Behavioural interventions that decrease HPA axis overactivation
Conclusion: Mitochondria are central regulators that play a predominant role in shaping the biological response to stress.
This paper was one of the earliest authoritative statements to position mitochondria at the crossroads of stress adaptation, mental health, and disease. According to the authors, maintaining mitochondrial integrity is essential for buffering, balanced stress responses, and the breakdown of mitochondrial control mechanisms marks a key transition from adaptive stress to chronic pathology.
What sets this 2007 paper apart is that it predicted much of what later research (e.g., Picard, McEwen, Daniels) confirmed, shaping research in psychiatry, neuroendocrinology, and molecular medicine, specifically:
• The dual (adaptive/maladaptive) role mitochondria play in stress
• Their centrality to HPA axis feedback
• Their influence on glucocorticoid sensitivity
• The long-term effects of early stress exposure on mitochondrial capacity
___
Manoli, I. Alesci, S. Blackman, MR. et al. (2007). Mitochondria as key components of the stress response. Trends in Endocrinology & Metabolism. 18(5), pp. 190-198. doi: 10.1016/j.tem.2007.04.004
“Under normal physiological conditions, reactive oxygen species (ROS) serve as ‘redox messengers’ in the regulation of intracellular signalling, whereas excess ROS may induce irreversible damage to cellular components and lead to cell death by promoting the intrinsic apoptotic pathway through mitochondria. In the aging process, accumulation of mitochondria DNA mutations, impairment of oxidative phosphorylation as well as an imbalance in the expression of antioxidant enzymes result in further overproduction of ROS. This mitochondrial dysfunction-elicited ROS production axis forms a vicious cycle, which is the basis of mitochondrial free radical theory of aging. In addition, several lines of evidence have emerged recently to demonstrate that ROS play crucial roles in the regulation of cellular metabolism, antioxidant defence and posttranslational modification of proteins. We first discuss the oxidative stress responses, including metabolites redistribution and alteration of the acetylation status of proteins, in human cells with mitochondrial dysfunction and in aging. On the other hand, autophagy and mitophagy eliminate defective mitochondria and serve as a scavenger and apoptosis defender of cells in response to oxidative stress during aging. These scenarios mediate the restoration or adaptation of cells to respond to aging and age-related disorders for survival. In the natural course of aging, the homeostasis in the network of oxidative stress responses is disturbed by a progressive increase in the intracellular level of the ROS generated by defective mitochondria. Caloric restriction, which is generally thought to promote longevity, has been reported to enhance the efficiency of this network and provide multiple benefits to tissue cells. ”
The Science-Based Strategies to Reboot Your Energy From the Inside Out
Fortunately, mitochondrial health is highly responsive to lifestyle change. You have more control than you think. Below are the most evidence-based tools to restore mitochondrial performance and reduce the cellular cost of stress:
1. Prioritise High-Quality Sleep
• Keep a consistent sleep schedule—even on weekends
• Limit blue light exposure 1–2 hours before bed
• Maintain a cool, quiet, and dark sleep environment
• Time caffeine consumption to mornings only
Sleep is when mitochondria undergo repair, clean-up (via mitophagy), and replication. Shortening or fragmenting sleep interferes with all of these processes.
Clinical correlation: Disrupted circadian rhythm and cortisol rhythms have been directly linked to reduced mitochondrial efficiency (Cell Reports, 2021).
2. Incorporate Mitochondria-Friendly Movement
Regular aerobic movement increases mitochondrial biogenesis. Just 20–30 minutes a day of brisk walking, cycling, jogging, or swimming can:
Increase enzyme activity in mitochondria
Improve oxygen delivery/ATP production
Reduce oxidative stress over time
Bonus tip: Incorporate short bursts of high-intensity intervals (HIIT) once or twice a week to enhance mitochondrial resilience.
3. Eat an Antioxidant-Dense, Anti-Inflammatory Diet
Key foods to support mitochondrial function include:
Polyphenols (berries, green tea, dark chocolate, and red grapes)
Sulfur compounds (garlic, onions, brassicas, including sprouted purple broccoli and kale, and Brussels sprouts)
Omega-3 fatty acids (wild salmon, sardines, and flax/linseeds)
Spices (turmeric, ginger, and rosemary)
• Coenzyme Q10-rich foods (organ meats, oily fish, and spinach)
• Magnesium & B vitamins (nuts, seeds, avocado, brassicas)
Including these in your diet fights oxidative stress, supports ATP production, and reduces inflammation that impairs mitochondrial function. You may also add intermittent fasting to your routine. An easily implementable fasting routine could be the 16-8, where you consume your regular calorie intake within 8 hours and increase your overnight fast to 16 hours.
4. Manage Your Mental Stress Load
Mind-body practices can reset your HPA axis, reduce cortisol, and lessen the strain on mitochondria:
Mindfulness meditation (10–20 minutes/day)
Breathwork (box breathing, 4-7-8 technique), as often as possible
Journaling (especially gratitude-based)
Nature walks (forest bathing), gardening, or earthing (barefoot walking outdoors)
Limit constant digital stimulation and information overload
Clinical trials have shown that mindfulness-based stress reduction effectively reduces pro-inflammatory gene expression and improves mitochondrial function.
5. Targeted Supplementation for Mitochondrial Support
Top mitochondrial supports:
• Coenzyme Q10 — to support the electron transport chain and ATP production
• Acetyl-L-carnitine — helps transport fatty acids into mitochondria for energy
• PQQ (Pyrroloquinoline quinone) — stimulates mitochondrial biogenesis
• Alpha-lipoic acid — an antioxidant that protects mitochondria from damage
• Magnesium — needed for hundreds of mitochondrial functions
• NAC (N-acetylcysteine) — supports intracellular glutathione, a powerful antioxidant
• Rhodiola rosea, holy basil (Tulsi), ashwagandha and medicinal mushrooms — adaptogens shown to modulate cortisol and support stress resilience
Note: Always consult a healthcare professional before taking any supplements. Ensure that they are from trusted sources and therapeutic grade.
Mitochondria: The Bridge Between Stress and Energy
At a glance, fatigue, brain fog, mood swings, and low resilience may seem unrelated, but beneath the surface, your mitochondria may be the cause, silently sapping your energy. Yet the story doesn’t end there.
Modern research agrees: Lifestyle change isn’t just preventative, it’s regenerative, especially when it comes to mitochondrial health. Real recovery starts in the cell. Energy isn’t just something you get; it’s something your body makes. And you have incredible power to influence how much it produces.
Remember that chronic stress impairs mitochondrial health by hindering the formation of new mitochondria (reduced biogenesis), preventing the removal of damaged mitochondria (impaired mitophagy), increasing the production of ROS (oxidative stress), and promoting low-grade inflammation that further burdens cellular energy production. All of these processes result in lower ATP production, and your energy levels decrease, even with sufficient sleep and a healthy diet.
Reclaim Your Energy Today
If any of the above signs of mitochondrial stress apply to you, take it as a call to pause, not to panic.
Manage stress and anxiety through mindful rituals.
Fuel your mitochondria with the right food and movement. Add intermittent fasting to your routine (for example, 16-8 extended overnight fast once or twice a week)
Let sleep become your top biohack.
And most importantly, remember: your burnout is not just in your head; it started in your cells.
Start small, stay consistent, and watch your energy return from the inside out.
Your body can’t perform better unless your cells do. Protect your mitochondria, and they’ll empower your mind, mood, and movement for life.
— Remember: Energy starts in your cells —
The Next Step:
Curious about mitochondrial health and want to get more energy?
Sign up for The 30-day Reset, a scientifically designed programme to reset your gut, circadian rhythm, improve nutrition, and reduce cellular stress, one day at a time.
The next session will start on August 18th.
References
Allen, J. Caruncho, HJ. Kalynchuk, LE. (2021). Severe life stress, mitochondrial dysfunction, and depressive behavior: A pathophysiological and therapeutic perspective. Mitochondrion. 56, pp. 111-117. doi: 10.1016/j.mito.2020.11.010
Anderson, EJ. Lustig, ME. Boyle, KE. et al. (2009). Mitochondrial H₂O₂ emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. Journal of Clinical Investigation. 119(3), pp. 573–581. doi:10.1172/JCI37048
Beal, MF. (2005). Mitochondria take center stage in aging and neurodegeneration. Annals of Neurology. 58(4), pp. 495-505. doi:10.1002/ana.20624
Choi, AM. Ryter, SW. Levine, B. (2013). Autophagy in human health and disease. New England Journal of Medicine. 368(7), pp. 651–662. doi:10.1056/NEJMra1205406
Clostre, F. (2001). Mitochondria: Recent pathophysiological discoveries and new therapeutic perspectives. [French]. Annales Pharmaceutiques Françaises. 59(1), pp. 3-21.
Daniels, TE. Olsen, EM. Tyrka, AR. (2020). Stress and psychiatric disorders: The role of mitochondria. Annual Reviews in Clinical Psychology. 16, pp. 165-186. doi:10.1146/annurev-clinpsy-082719-104030
Du, J. Wang, Y. Hunter, R. et al. (2009). Dynamic regulation of mitochondrial function by glucocorticoids. Proceedings of the National Academy of Sciences of the United States of America. 106(9), pp. 3543-3548. doi:10.1073/pnas.0812671106
Duchen, MR. (2004). Mitochondria in health and disease: Perspectives on a new mitochondrial biology. Molecular Aspects of Medicine. 25(4), pp. 365-451. doi:10.1016/j.mam.2004.03.001
Fajardo, ME. Phillips, SM. (2019). Oxidative stress, mitochondrial function, and aging: Therapeutic considerations. Journal of Gerontology: Biological Sciences. 74(3), pp. 185–194. doi:10.1093/gerona/gly055
Harris, RB. (2015). Chronic and acute effects of stress on energy balance: Are there appropriate animal models? American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 308(4), R250–R265. doi:10.1152/ajpregu.00361.2014
Higgins, GC. Beart, PM. Shin, YS. et al. (2010). Oxidative stress: Emerging mitochondrial and cellular themes and variations in neuronal injury. Journal of Alzheimer’s Disease. 20(Suppl. 2), S453-S473. doi:10.3233/JAD-2010-100321
Hood, MS. Little, JP. Tarnopolsky, MA. et al. (2011). Low-volume interval training improves muscle oxidative capacity in sedentary adults. Medicine & Science in Sports & Exercise. 43(10), pp. 1849–1856. doi:10.1249/MSS.0b013e3182199834
Ip, CK. Herzog, H. Zhang, L. (2019). Chronic stress and energy homeostasis. Aging (Albany NY). 11(22), pp. 9963–9964. doi:10.18632/aging.102500
Kazantsev, AG. (2007). Cellular pathways leading to neuronal dysfunction and degeneration. Drug News & Perspectives. 20(8), pp. 501-509. doi:10.1358/dnp.2007.20.8.1157616
Kumar, A. Hood, DA. (2015). Regulation of endogenous antioxidants by physical activity in muscle mitochondrial biogenesis. Antioxidants & Redox Signaling. 22(4), pp. 768–783. doi.:10.1089/ars.2014.5852
Kwong, JQ. Beal, MF. Manfredi, G. (2006). The role of mitochondria in inherited neurodegenerative diseases. Journal of Neurochemistry. 97(6), pp. 1659-1675. doi:10.1111/j.1471-4159.2006.03990.x
Lartigue, L. Faustin, B. (2013). Mitochondria: Metabolic regulators of innate immune responses to pathogens and cell stress. The International Journal of Biochemistry & Cell Biology. 45(9), pp. 2052-2056. doi:10.1016/j.biocel.2013.06.014
Lin, MT. Beal, MF. (2006). Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 443(7113), pp. 787-795. doi:10.1038/nature05292
Manoli, I. Alesci, S. Blackman, MR. et al. (2007). Mitochondria as key components of the stress response. Trends in Endocrinology & Metabolism. 18(5), pp. 190-198. doi: 10.1016/j.tem.2007.04.004
Patten, DA. Germain, M. Kelly, MA. et al. (2010). Reactive oxygen species: Stuck in the middle of neurodegeneration. Journal of Alzheimer’s Disease. 20(Suppl. 2), S357-S367. doi:10.3233/JAD-2010-100498
Picard, M. McEwen, BS. (2018). Psychological stress and mitochondria: A systematic review. Psychosomatic Medicine. 80(2), pp. 141-153. doi:10.1097/PSY.0000000000000545
Picard, M. McEwen, BS. (2018). Psychological stress and mitochondria: A conceptual framework. Psychosomatic Medicine. 80(2), pp. 126-140. doi:10.1097/PSY.0000000000000544
Radak, Z. Zhao, Z. Koltai, E. et al. (2013). Oxygen consumption and usage during physical exercise: The balance between oxidative stress and ROS-dependent adaptive signaling. Antioxidants & Redox Signaling, 18(10), 1208–1246. doi:10.1089/ars.2011.4458
Rahman, MM. Tumpa, MAA. Rahaman, MS. et al. (2023). Emerging promise of therapeutic approaches targeting mitochondria in neurodegenerative disorders. Current Neuropharmacology. 21(5), pp. 1081-1099. doi:10.2174/1570159X21666230316150559
Rami, A. (2009). Review: Autophagy in neurodegeneration: Firefighter and/or incendiarist? Neuropathology and Applied Neurobiology. 35(5), pp. 449-461. doi:10.1111/j.1365-2990.2009.01034.x
Reid, DM. Choe, JY. Bruce, MA. et al, (2025). Mitochondrial functioning: Front and center in defining psychosomatic mechanisms of allostasis in health and disease. Methods in Molecular Biology. 2868, pp. 91-110. doi:10.1007/978-1-0716-4200-9_6
Sas, K. Robotka, H. Toldi, J. et al. (2007). Mitochondria, metabolic disturbances, oxidative stress and the kynurenine system, with focus on neurodegenerative disorders. Journal of the Neurological Sciences. 257(1-2), pp. 221-239. doi:10.1016/j.jns.2007.01.033
Trushina, E, McMurray, CT. (2007). Oxidative stress and mitochondrial dysfunction in neurodegenerative diseases. Neuroscience. 145(4), pp. 1233-1248. doi: 10.1016/j.neuroscience.2006.10.056
van der Kooij, MA. (2020). The impact of chronic stress on energy metabolism. Molecular and Cellular Neuroscience. 107, 103525. doi:10.1016/j.mcn.2020.103525
Wang, CH. Wu, SB. Wu, YT. et al. (2013). Oxidative stress response elicited by mitochondrial dysfunction: Implication in the pathophysiology of aging. Experimental Biology and Medicine (Maywood). 238(5), pp. 450-460. doi:10.1177/1535370213493069