The liver–brain connection: Its Role in Inflammation, Mental Health and Chronic Disease
The liver–brain connection is a two‑way communication network linking metabolism, detoxification, immunity and cognition, primarily via the gut–liver–brain axis. When this system is balanced, the liver helps stabilise blood sugar, hormones, toxin removal, and inflammatory signals, so the brain can think clearly, regulate mood, and maintain a healthy stress response. When it is disrupted, we see everything from brain fog and low mood to severe liver issues and increased dementia risk.
How the liver and brain talk to each other
Communication runs through several intertwined channels:
Neural pathways:
The vagus and sympathetic nerves carry signals between the liver, gut and brain, allowing the brain to adjust liver glucose output, bile flow and immune activity, and the liver to feed back information on energy status and inflammation.
Blood‑borne signals:
The liver regulates glucose, lipids, ketones, and amino acids, and detoxifies xenobiotics; shifts in these metabolites directly influence brain energy supply, neurotransmitter synthesis, and oxidative stress.
Immune and inflammatory mediators:
Cytokines and acute‑phase proteins produced in the liver (and gut) can cross the blood–brain barrier (or signal across it), shaping microglial activation, neuroinflammation, and neuronal health.
Gut microbiota and their metabolites:
Microbial products (short‑chain fatty acids, bile‑acid metabolites, tryptophan derivatives, endotoxin) travel via the portal vein to the liver, then into systemic circulation, influencing both hepatic function and brain signalling.
Together, these pathways form what is now often described as the gut–liver–brain axis.
What disturbs the liver–brain connection?
Several common patterns can destabilise this axis. They often coexist and amplify each other.
1. Chronic liver disease and ammonia overload
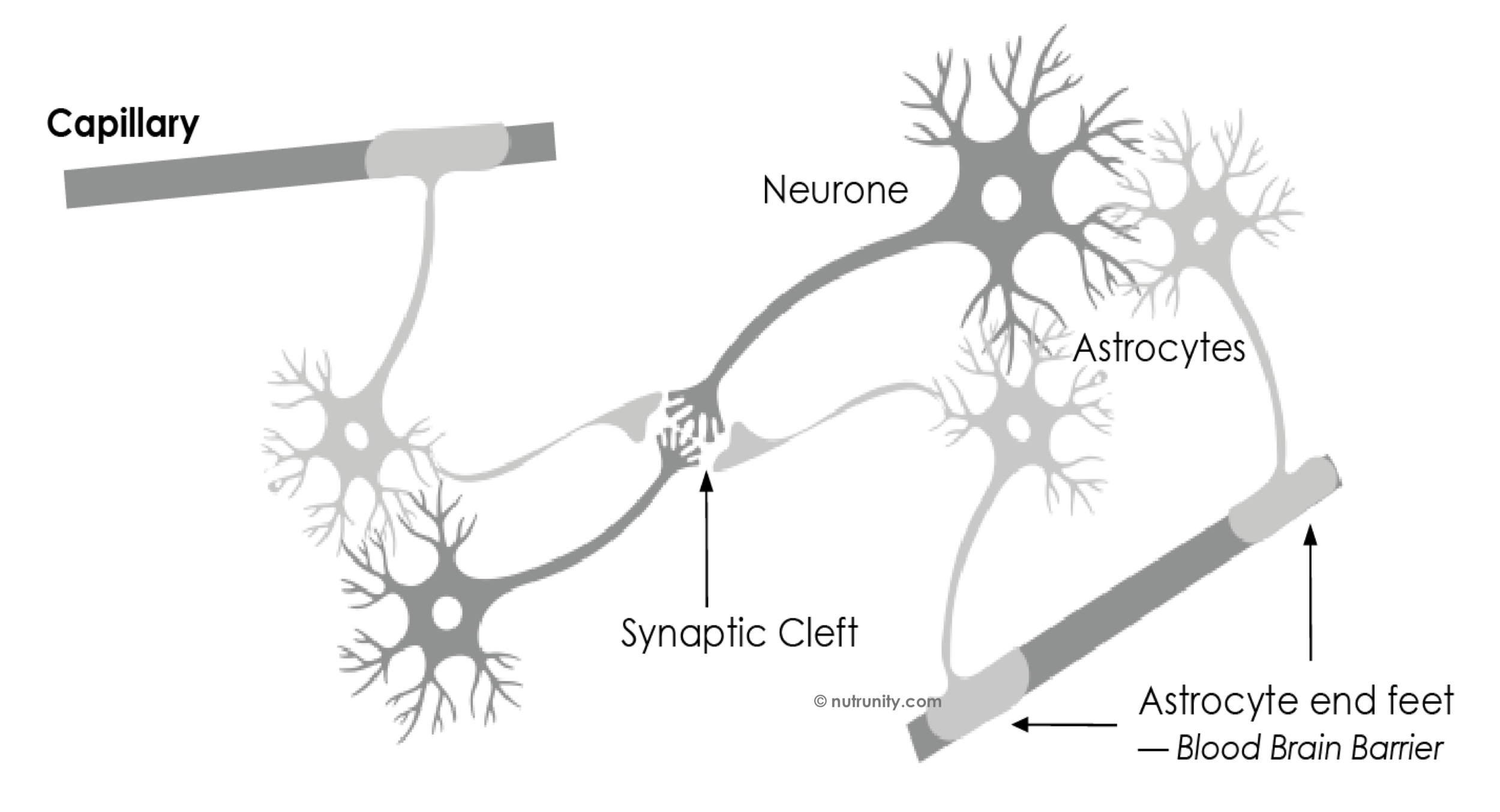
In advanced liver disease or cirrhosis, the liver’s ability to detoxify ammonia and other nitrogenous waste falls. Ammonia and related metabolites can then accumulate, cross into the brain and disrupt astrocyte and neuron function, contributing to hepatic encephalopathy (HE), a spectrum from subtle cognitive changes to confusion, coma and death.
Research shows that a non-functioning liver can lead to death in just 24 hours. The liver's role in eliminating toxins and unwanted substances is that important.
Mechanisms include:
Astrocyte swelling and brain oedema.
Altered glutamate/GABA balance and increased inhibitory tone.
Oxidative and nitrosative stress and impaired mitochondrial function in brain cells.
This is the most dramatic example of impaired liver–brain crosstalk.
Astrocyte with direct access to blood vessels
Illustration extracted from Energise - 30 Days to Vitality. all rights reserved.
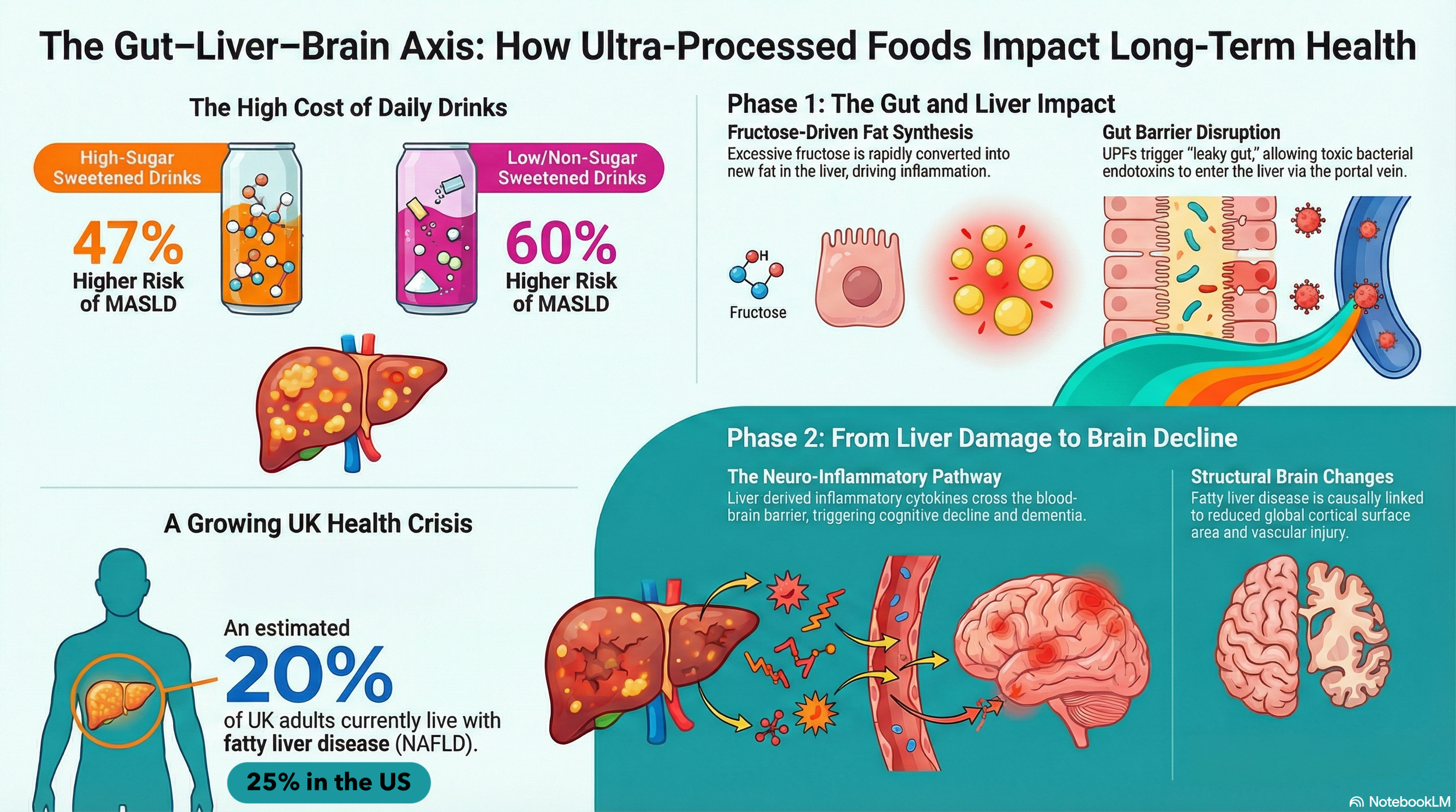
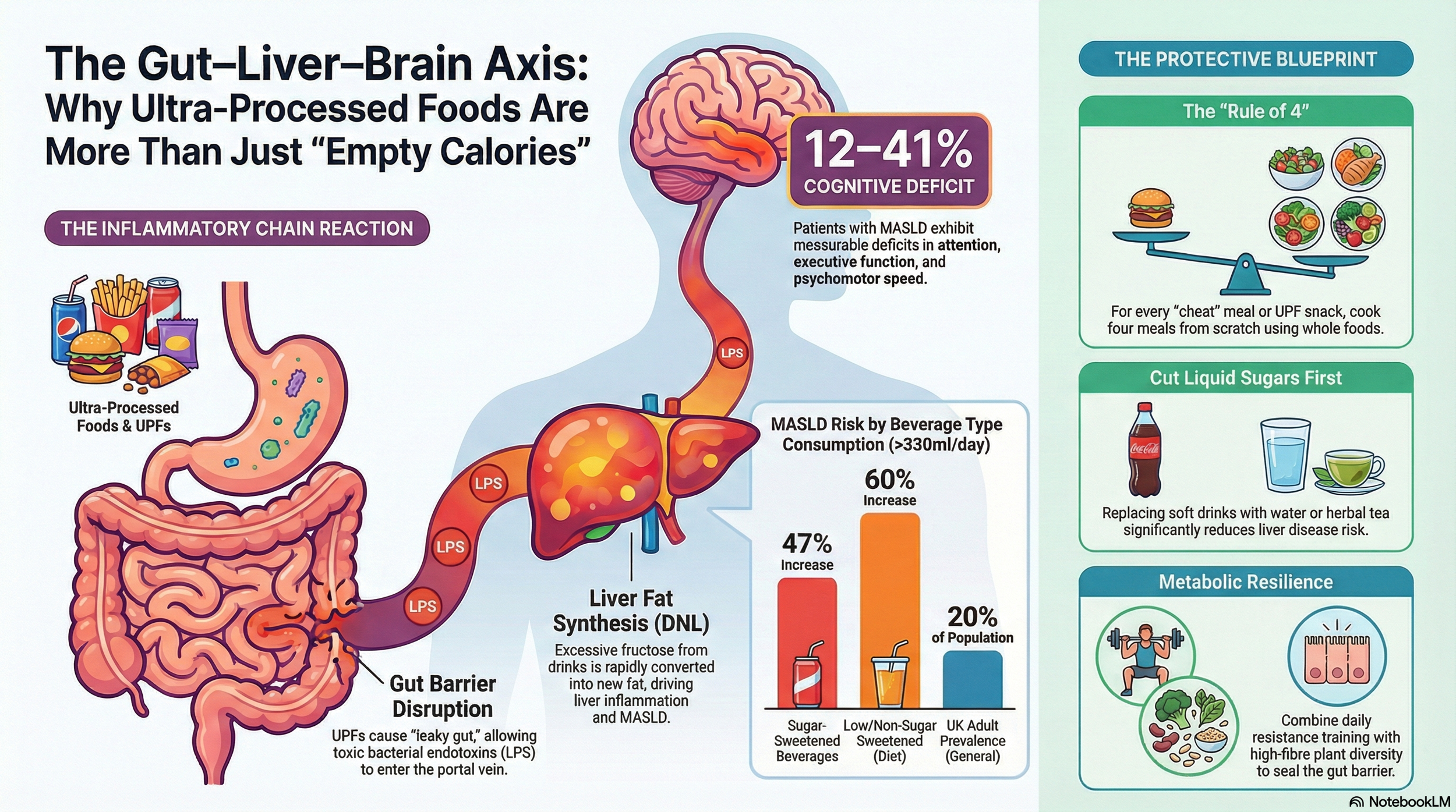
2. Non‑alcoholic fatty liver disease (NAFLD) and metabolic syndrome
NAFLD and metabolic syndrome have a widespread effect. Fatty, inflamed liver tissue releases pro‑inflammatory cytokines and alters lipid and glucose handling, which is associated with cognitive impairment and higher dementia risk, especially in mid‑life.
Cohort studies show:
NAFLD is independently associated with poorer learning, recall and concentration.
Mid‑life NAFLD roughly triples the risk of later cognitive impairment in some populations, likely via vascular and inflammatory pathways.
3. Gut dysbiosis and increased intestinal permeability
Disruption of the gut microbiota can increase intestinal permeability, allowing endotoxins (LPS) and other microbial products, as well as problematic proteins, allergens, and environmental pollutants, into the portal circulation. The liver responds with inflammation and changes in bile acids and metabolites, which, in turn, affect systemic immunity and the brain.
In cirrhosis and HE, specific microbiota changes (overgrowth of urease‑producing and ammonia‑generating bacteria) worsen hyperammonaemia and neuroinflammation. Even in the absence of overt liver disease, dysbiosis is linked to low‑grade systemic inflammation and altered neurotransmitter pathways along the gut–brain axis.
4. Systemic inflammation and blood–brain barrier disruption
Persistent inflammatory signalling from the liver and gut can alter the blood–brain barrier (BBB) integrity and activate microglia. Over time, this can contribute to neurodegenerative processes and mood disorders by:
Increasing oxidative stress in neurons.
Impairing the clearance of toxic proteins such as beta‑amyloid.
Altering synaptic plasticity and neurotransmission.
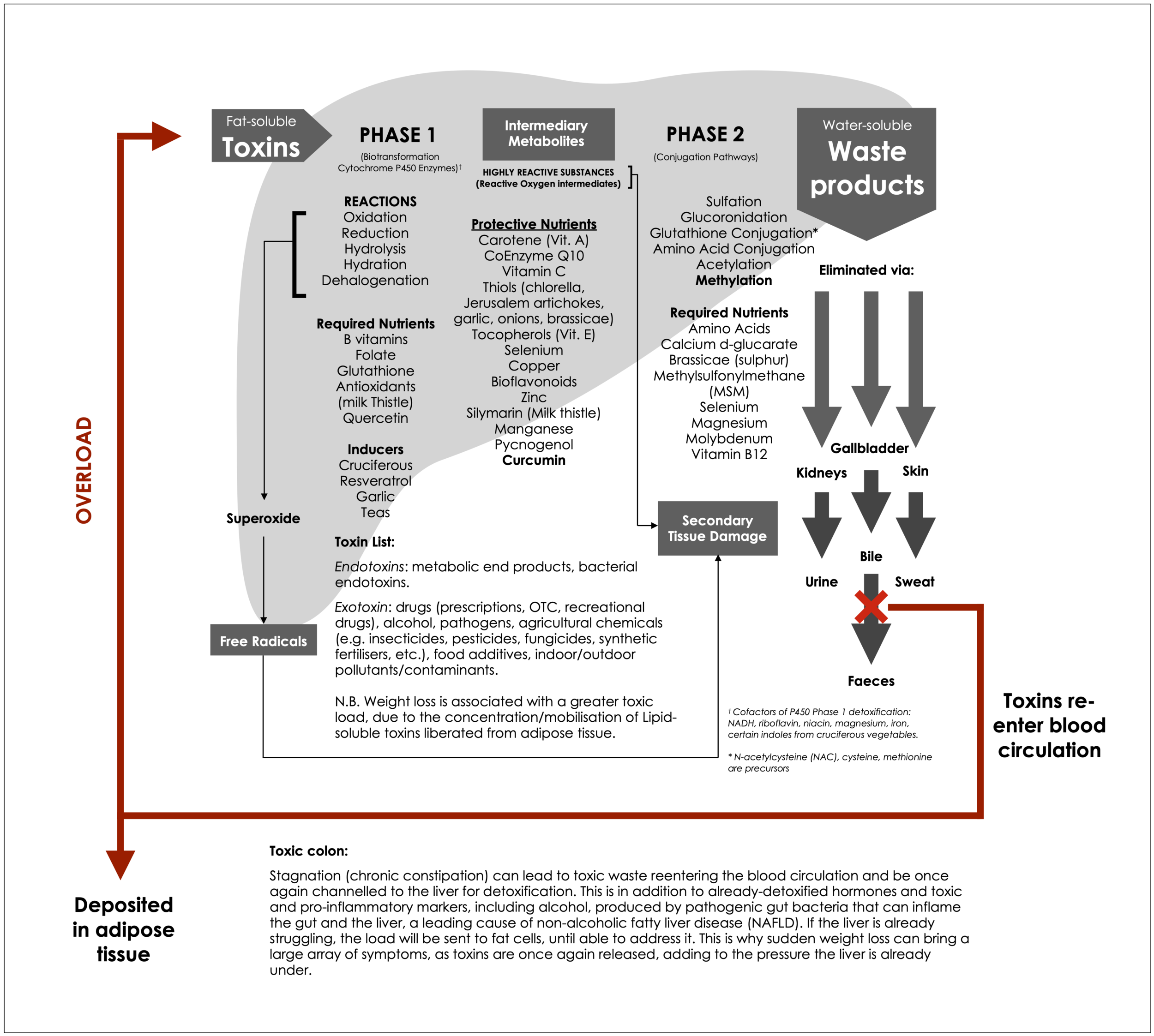
5. Toxins, alcohol and medications
The liver is the main site for detoxifying alcohol, drugs and environmental toxins. Chronic exposure can:
Damage hepatocytes (steatosis, fibrosis, cirrhosis).
Generate reactive metabolites and oxidative stress.
Increase circulating neurotoxic substances when clearance is compromised.
Alcohol‑related liver disease is a major risk factor for HE and broader cognitive decline. Certain medications can also worsen HE in susceptible patients by affecting ammonia production, constipation, renal function or CNS sensitivity.
Poor liver function can also have severe consequences in the body. This can be provoked by excessive exposure to pollutants and toxicants (think living near a highway, indoor pollution, and toxic chemicals in the home), low nutrient intake, and high consumption of ultra-processed manufactured food products (packed with health-wrecking additives, chemical formulations, and ultra-refined ingredients). stressful lifestyle, poor sleep, and chronic constipation, among other factors.
6. Brain‑first insults feeding back to the liver and gut
The traffic is not one‑way. Brain disorders (stroke, trauma, chronic stress, depression) can alter autonomic output to the liver and gut, impacting motility, bile flow, barrier function and microbial composition. This can worsen metabolic control and inflammation, creating a vicious circle across the liver–brain axis.
What helps restore a healthier liver–brain connection?
Restoration focuses on protecting the liver, calming systemic and neuro‑inflammation, normalising gut ecology and supporting metabolic control, with some interventions specific to advanced disease.
1. Treating underlying liver disease
For cirrhosis and HE, core medical measures include:
Reducing ammonia production and absorption (e.g. non‑absorbable disaccharides such as lactulose, and non‑absorbable antibiotics such as rifaximin).
Managing precipitating factors (constipation, infection (including gut dysbiosis), GI bleeding, electrolyte imbalance). These strategies lower neurotoxic load and can partially reverse cognitive symptoms by restoring cleaner liver–brain signalling.
In NAFLD and NASH, weight reduction and improved glycaemic control can reduce liver fat and inflammation, with evidence for parallel improvement in cognitive outcomes.
2. Improving metabolic health
Because metabolic dysfunction sits at the centre of many liver–brain problems, interventions that improve insulin sensitivity, lipid profiles and blood pressure support both organs. These include:
Dietary patterns rich in whole, minimally processed foods, high in fibre and unsaturated fats (e.g. Mediterranean‑style diets).
Regular physical activity, which improves hepatic and peripheral insulin sensitivity and enhances brain‑derived neurotrophic factor (BDNF) and cerebral blood flow.
Addressing sleep apnoea and circadian disruption, both of which worsen insulin resistance, NAFLD and cognitive health (particularly via the stress response and hypercortisolaemia).
3. Targeting the gut microbiota
Modulating the microbiota–gut–liver–brain axis is an active area of research. Approaches include:
Dietary fibre and prebiotics, which increase SCFA production and support barrier integrity and anti‑inflammatory signalling.
Probiotics or synbiotics, which have shown modest benefits in some studies for reducing HE episodes and improving minimal HE, likely do so by reducing ammonia‑producing bacteria and improving gut barrier function.
Emerging therapies, such as faecal microbiota transplantation, for refractory HE are still under investigation.
By reducing endotoxaemia and improving metabolite profiles, these strategies can indirectly support both liver and brain function.
4. Supporting barrier integrity (gut and brain)
Interventions that stabilise intestinal and blood–brain barriers help limit toxin and cytokine exposure to the CNS. These include:
Anti‑inflammatory diets, rich in polyphenols, prebiotic fibres, and omega‑3 fatty acids.
Optimising vitamin D, B‑vitamin and antioxidant status, which are important for tight‑junction proteins and redox balance.
Managing conditions that damage barriers (chronic stress, high alcohol intake, poorly controlled hypertension and diabetes). This also includes mindful eating and eating at the table as a family.
5. Reducing toxic load (alcohol and environmental exposures)
Limiting alcohol and hepatotoxic medications, avoiding unnecessary polypharmacy, and reducing exposure to environmental toxins that burden hepatic detoxification all decrease the risk of liver injury and its knock‑on brain effects. This is particularly important in people with existing liver impairment.
6. Nervous‑system regulation and stress reduction
Chronic psychological stress alters autonomic balance (sympathetic/vagal) and HPA‑axis function, influencing hepatic glucose output, gut motility, microbiota composition and inflammatory tone. Stress‑reduction practices, such as mindfulness, breathing practices, yoga and appropriate psychotherapy, can improve vagal tone and reduce systemic inflammation, with benefits across the gut–liver–brain axis.
7. Neuroprotective strategies
For those with established liver disease or metabolic risk, additional neuroprotective measures may help, such as:
Managing cardiovascular risk factors aggressively (blood pressure, lipids, smoking, excessive alcohol consumption) to reduce vascular contributions to cognitive decline in NAFLD.
Ensuring adequate cognitive stimulation, social engagement and physical activity to build cognitive reserve.
Putting it together
The liver–brain connection is a network that includes gut microbes, intestinal barriers, immune signalling, autonomic nerves and metabolic control. It can be disrupted by advanced liver disease, fatty liver, dysbiosis, systemic inflammation, toxins, and even primary brain disorders that feed back to the liver and gut.
Restoring this axis means working on multiple fronts: treating liver disease when present, improving metabolic health, reshaping the gut microbiota, supporting gut and brain barriers, reducing the toxic burden, and regulating the nervous system. Taken together, these strategies help the liver resume its role as a metabolic and detoxification hub and allow the brain to operate with a cleaner, more stable internal environment.
References:
Bartholdy, A., Moseholm, KF., Nielsen, PY. et al. (2026). Long-term dementia risk in metabolic dysfunction-associated steatotic liver disease: a population-based study. Metabolic Brain Disease. 41(1), 2026. doi:10.1007/s11011-026-01796-x
Costa, CFFA., Ferreira-Gomes, J., Barbosa, F. et al. (2024). Importance of good hosting: reviewing the bi-directionality of the microbiome-gut-brain-axis. Frontiers in Neuroscience. 18, 1386866. doi:10.3389/fnins.2024.1386866
Cushman, M., Callas, PW., Alexander, KS. et al. (2023). Nonalcoholic fatty liver disease and cognitive impairment: A prospective cohort study. PLoS One. 18(4), e0282633. doi:10.1371/journal.pone.0282633
Jayakumar, AR., Rama Rao, KV., Norenberg, MD. (2015). Neuroinflammation in hepatic encephalopathy: Mechanistic aspects. Journal of Clinical and Experimental Hepatology. 5(Suppl 1), S21-8. doi:10.1016/j.jceh.2014.07.006
Lu, H., Zhang, H., Wu, Z. et al. (2024). Microbiota-gut-liver-brain axis and hepatic encephalopathy. Microbiome Research Reports. 3(2), 17. doi:10.20517/mrr.2023.44
Matsubara, Y., Kiyohara, H., Teratani, T. et al. (2022). Organ and brain crosstalk: The liver-brain axis in gastrointestinal, liver, and pancreatic diseases. Neuropharmacology. 205, 108915. doi:10.1016/j.neuropharm.2021.108915
Pan, L., Xie, L., Yang, W. et al. (2025). The role of brain-liver-gut axis in neurological disorders. Burns & Trauma. 13, tkaf011. doi:10.1093/burnst/tkaf011
Seo, SW., Gottesman, RF., Clark, JM. et al. (2016). Nonalcoholic fatty liver disease is associated with cognitive function in adults. Neurology. 86(12), pp. 1136-1142. doi:10.1212/WNL.0000000000002498
Sørensen, M., Andersen, JV., Bjerring, PN. et al. (2024). Hepatic encephalopathy as a result of ammonia-induced increase in GABAergic tone with secondary reduced brain energy metabolism. Metabolic Brain Disease. 40(1), 19. doi:10.1007/s11011-024-01473-x
Sun, X., Shukla, M., Wang, W. et al. (2024). Unlocking gut-liver-brain axis communication metabolites: Energy metabolism, immunity and barriers. NPJ Biofilms Microbiomes. 10(1), 136. doi:10.1038/s41522-024-00610-9
Yan, M., Man, S., Sun, B. et al. (2023). Gut liver brain axis in diseases: The implications for therapeutic interventions. Signal Transduction and Targeted Therapy. 8(1), 443. doi:10.1038/s41392-023-01673-4
Wu, H., Zhang, Y., Yu, J. et al. (2023). Editorial: Gut-liver-brain axis: A complex network influences human health and diseases. Frontiers in Neuroscience. 17, 1241069. doi:10.3389/fnins.2023.1241069