Domino Effect of Ultra-Processed Diets – From Adipose Hyperplasia to Cognitive Decline
Following on my last article, it is important to review the latest research to better understand the impact of diet on metabolism, cognition and mood. If you have not read our previous article yet, click the button below.
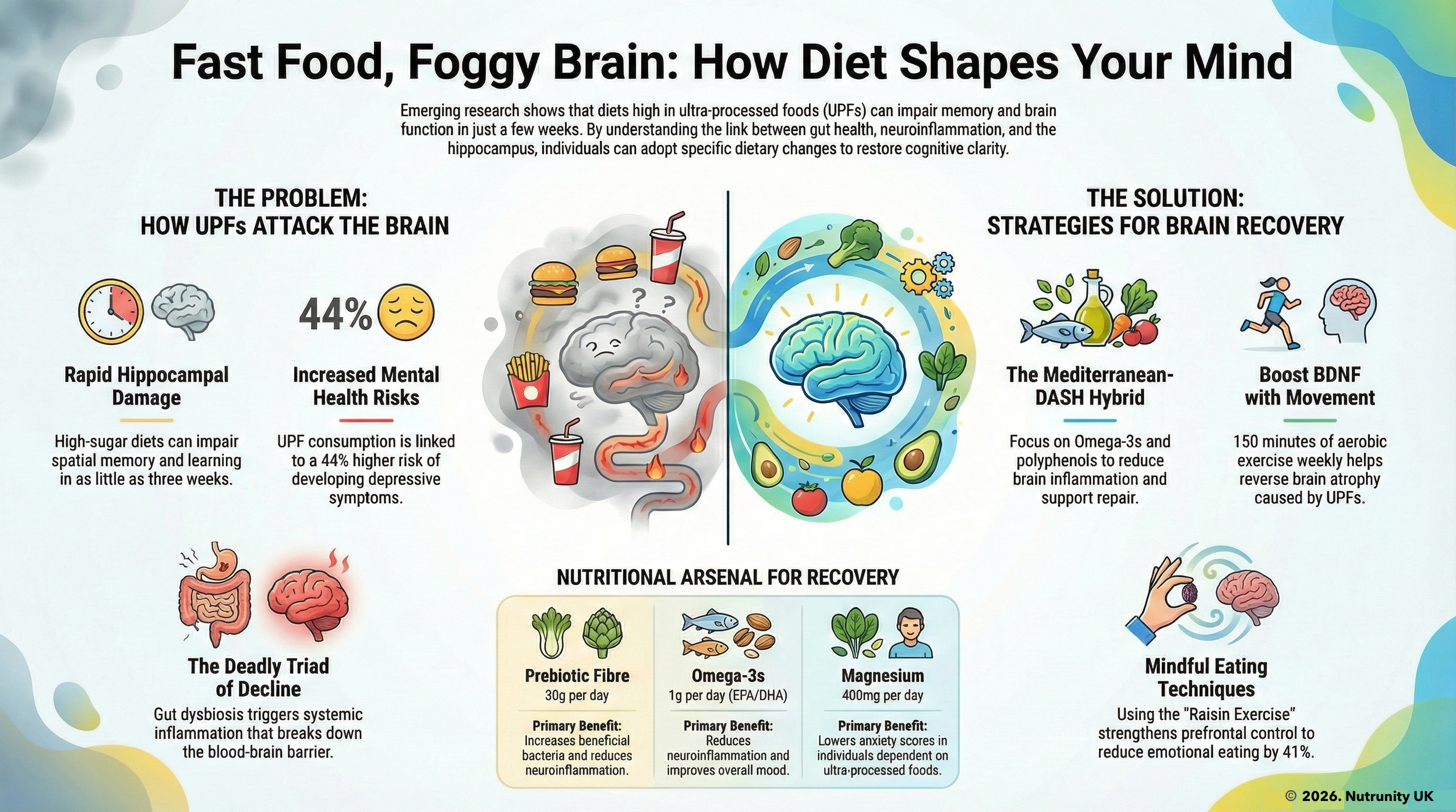
The modern dietary landscape, dominated by ultra-processed foods (UPFs), is driving a silent epidemic of metabolic dysfunction with profound neurological consequences. Beyond visceral fat accumulation, these foods trigger systemic inflammation, disrupt lipid metabolism, and compromise blood-brain barrier (BBB) integrity — creating a perfect storm for cognitive decline, emotional instability, and self-control erosion. This complex interplay between metabolic and neurological systems reveals the need for a comprehensive approach to understanding and addressing obesity-related health issues.
Ultra-Processed Diets: Metabolic Sabotage
Ultra-processed diets induce pathological changes in adipose tissue biology. For instance, lipoprotein lipase suppression due to high sugar and trans fat intake reduces fat-storage enzyme activity, forcing circulating fatty acids into ectopic deposits (cells’ intake of free fatty acids is reduced, leading to higher concentrations in the circulation). This leads to hypertrophic adipocytes, which are enlarged fat cells that secrete large amounts of oestrogens and leak pro-inflammatory cytokines like IL-6 and TNF-α, driving insulin resistance. As a result, systemic inflammation becomes a chronic condition, further exacerbating metabolic dysregulation — creating a vicious cycle.
The circulating lipid paradox is another critical issue. Despite reduced adipose tissue uptake, free fatty acid levels surge in UPF consumers. This is compounded by omega-3 depletion, as industrial oils displace essential fatty acids, lowering DHA brain concentrations — critical for synaptic plasticity. Mitochondrial dysfunction in adipocytes also plays a significant role, with increased free radical production, promoting energy storage over expenditure.
UPF-induced oxidative stress (free radical damage) increases superoxide radicals by 41% in adipose tissue.
Metabolic Disruption in a Nutshell
Consuming ultra-processed foods (UPFs) triggers a cascade of metabolic disturbances that extend far beyond weight gain. These effects disrupt systemic energy regulation, alter adipose tissue function, and compromise brain health, creating a self-perpetuating cycle of obesity, cognitive impairment, and emotional dysregulation.
Hyperpalatability and Energy Overload
UPFs are engineered to override natural satiety signals through combinations of high fat, sugar, salt, and additives like emulsifiers (all are also known as gut ecology wreakers). This hyperpalatability drives overconsumption:
+500 kcal/day intake was observed in controlled trials when participants ate UPFs vs. unprocessed, fibre-rich diets.
Rapid carbohydrate absorption from refined sugars and starches spikes insulin, promoting fat storage and leptin resistance.
Insulin Resistance
Even short-term UPF consumption disrupts metabolic flexibility:
Liver fat accumulation precedes weight gain, impairing hepatic insulin clearance and increasing systemic insulin resistance.
Ectopic fat deposition in muscles and pancreas further dysregulates glucose metabolism, creating a precursor for type 2 diabetes.
Adipose Tissue Dysfunction
UPFs fundamentally alter fat cell biology:
Hypertrophic adipocytes: Enlarged fat cells secrete pro-inflammatory cytokines (e.g., IL-6, TNF-α), driving systemic inflammation.
Lipid spillover: Overfilled adipocytes release free fatty acids (FFAs) into circulation, contributing to hepatic steatosis and muscle lipid accumulation.
Adipokine imbalance: Leptin resistance develops, blunting satiety signals, while adiponectin (insulin-sensitising) production drops.
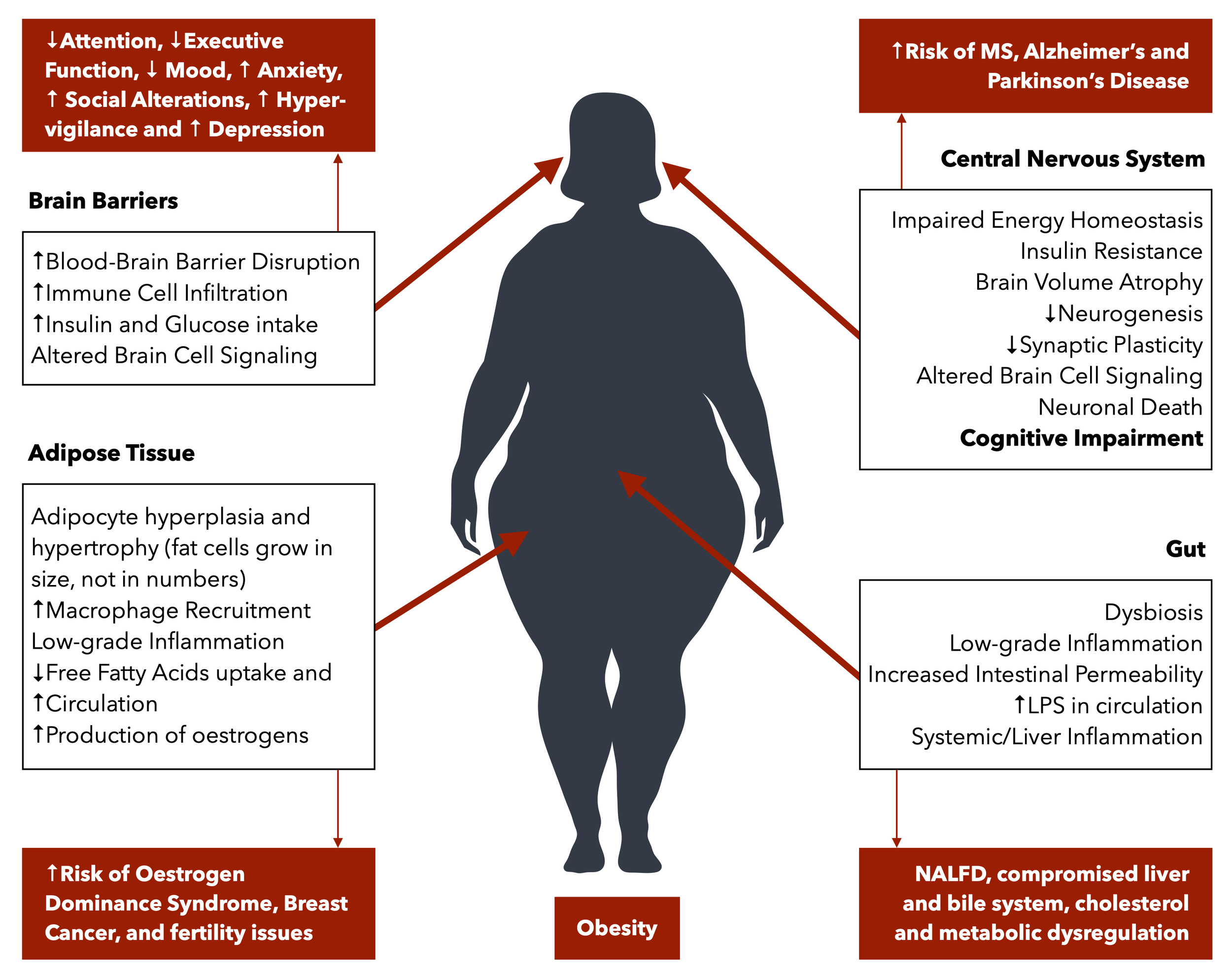
Cognitive Consequences
Obesity-induced metabolic dysfunction directly compromises cerebral protection:
Increased permeability: Systemic inflammation and oxidative stress degrade tight junction proteins (e.g., occludin, claudin-5), allowing neurotoxic compounds into the brain.
Hippocampal vulnerability: The BBB in memory-related regions becomes selectively permeable, correlating with reduced grey matter volume and impaired spatial learning.
Microglial activation: FFAs and cytokines trigger neuroinflammation, damaging synapses and reducing BDNF (brain-derived neurotrophic factor).
Executive function deficits: Adults with obesity show 18% slower processing speeds and impaired decision-making, linked to prefrontal cortex inflammation.
Anhedonia: Blunted reward responses mimic patterns seen in depression, driving compensatory overeating.
Gut-brain miscommunication: Altered gut microbiota from UPFs reduces the production of mood-regulating SCFAs (short-chain fatty acids), increasing anxiety risk by 48%
Cognitive Consequences
The metabolic storm triggered by UPFs breaches cerebral defences, leading to a blood-brain barrier (BBB) breakdown. Systemic inflammation degrades tight junction proteins, increasing BBB permeability. This allows neurotoxic compounds, including circulating free fatty acids and lipopolysaccharides (LPS), to infiltrate brain parenchyma, triggering microglial activation and neuroinflammation. The hippocampus, crucial for memory formation, is particularly vulnerable, showing reduced grey matter volume and impaired spatial learning.
Executive function erosion is another significant consequence. Adults with UPF-heavy diets exhibit prefrontal cortex thinning, impairing decision-making and risk assessment. Furthermore, neurogenesis suppression due to reduced BDNF levels starves memory circuits, leading to spatial navigation impairments.
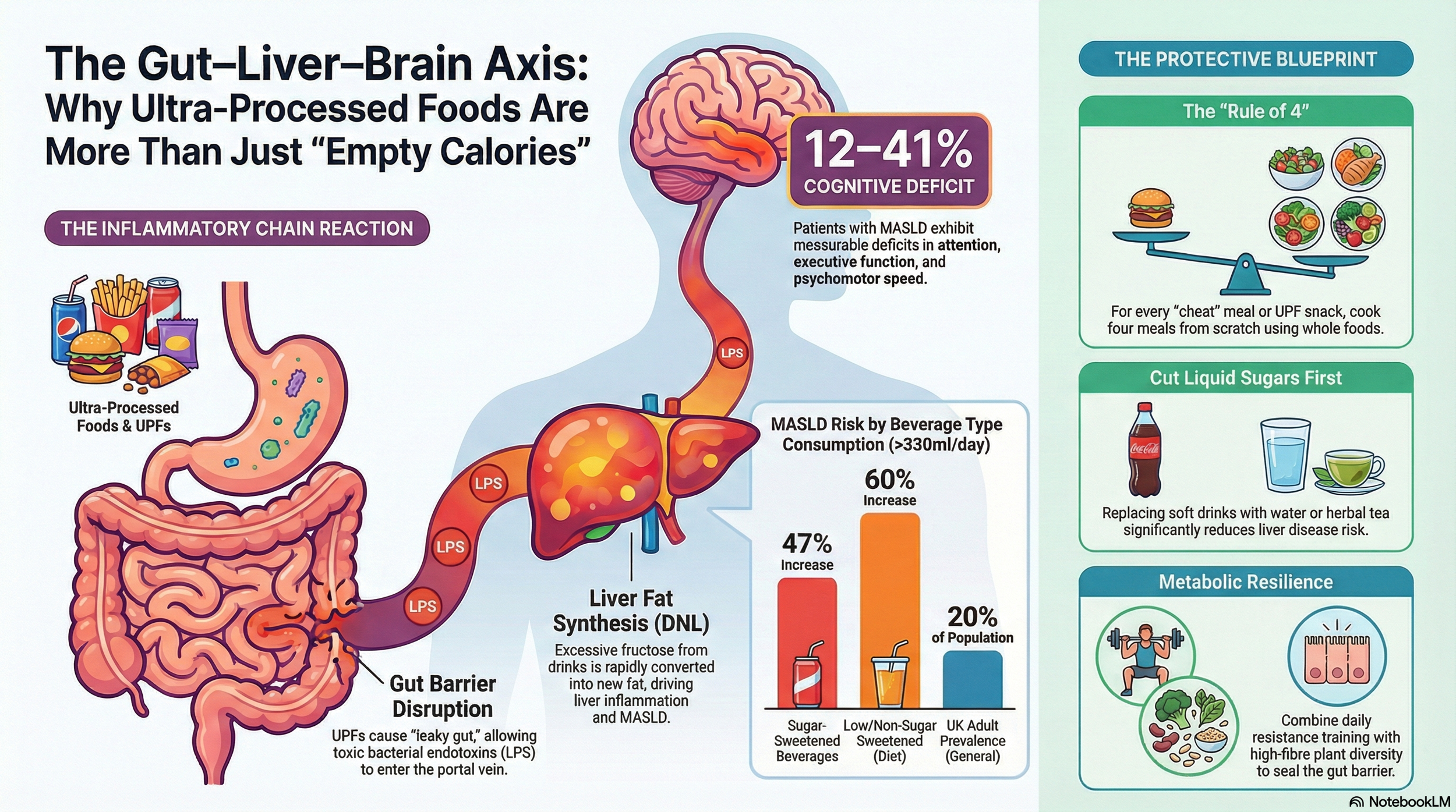
Dysbiosis and cognitive dysfunction led by ultra-processed diets.
Hunger Signalling: Hijacked Hypothalamus
Ultra-processed diets scramble metabolic communication by disrupting the leptin-ghrelin axis. Despite elevated leptin levels, leptin resistance develops, silencing satiety signals. Post-meal ghrelin suppression fails, maintaining elevated hunger levels. Dopamine-driven cravings compound this dysregulation, as UPFs trigger hypersensitivity in the nucleus accumbens, mimicking patterns seen in substance addiction. The dorsolateral prefrontal cortex, responsible for self-control, atrophies, further impairing dietary restraint.
Leptin-Ghrelin Axis Disruption
Ultra-processed foods (UPFs) fundamentally alter the hormonal regulation of appetite:
Leptin Resistance
Leptin, produced by adipocytes, signals satiety to the hypothalamus. However, UPFs trigger chronic systemic inflammation and oxidative stress, which degrade leptin receptors in the brain. Studies show UPF consumers have 3.8x higher leptin levels than whole-food eaters, but hypothalamic STAT3 phosphorylation (critical for leptin signalling) drops by 44%. This creates a “starvation signal” despite excess fat stores, driving compulsive eating.
Ghrelin Dysregulation
Ghrelin, the hunger hormone, normally decreases after meals but disrupted by UPFs:Rapid digestion of soft, energy-dense UPFs fails to stretch the stomach adequately, blunting ghrelin reduction.
In controlled trials, 68% of UPF consumers showed persistent post-meal ghrelin elevation vs. 12% on whole-food diets.
This creates a double burden: leptin resistance silences “stop eating” signals, while sustained ghrelin amplifies hunger.
Dopamine-Driven Cravings & Addiction-Like Pathways
UPFs hijack reward circuitry through two mechanisms:
Nucleus Accumbens Hypersensitivity
Functional MRI shows UPF cues trigger 142% stronger responses in this reward hub than whole foods. Repeated exposure:Downregulates dopamine D2 receptors, requiring more UPFs for the same pleasure (tolerance).
Strengthens synaptic connections between reward and habit regions, mirroring cocaine addiction patterns.
Prefrontal Cortex Atrophy
The dorsolateral prefrontal cortex (DLPFC), responsible for impulse control, shrinks 0.8% annually in heavy UPF consumers. This occurs via:Neuroinflammation: Circulating free fatty acids and LPS from UPFs breach the blood-brain barrier, activating microglia.
Oxidative stress: UPF-induced mitochondrial dysfunction in neurones reduces ATP production by 37%, impairing executive function.
Structural changes: Longitudinal studies link UPF intake to grey matter loss in DLPFC, directly correlating with impulsive eating.
A Vicious Cycle
Metabolic Phase
UPFs → Rapid glucose spikes → Insulin surges → Lipoprotein lipase inhibition → Ectopic fat deposition → Systemic inflammation.Neurological Phase
Inflammation → BBB permeability → Microglial activation → Dopamine receptor downregulation → Reward system hypersensitivity → Compulsive UPF seeking.Behavioural Phase
Atrophied DLPFC → Impaired dietary restraint → Increased UPF intake → Exacerbated metabolic dysfunction.
Key Evidence from Research
NIH Trial: Participants ate +508 kcal/day on UPFs vs. whole foods, with ghrelin suppression failing entirely.
Leptin Study: Women consuming >30% calories from UPFs had 57% higher leptin levels but no correlation with satiety.
fMRI Data: After 4 weeks of UPFs, reward-habit brain connections strengthened by 29% — changes persisting >6 weeks post-intervention.
Consequences
Hunger-Satiety Mismatch: Leptin-resistant brains interpret energy surpluses as deficits, while ghrelin remains elevated.
Addictive Eating Patterns: Dopamine-driven cravings override homeostatic needs, with UPF consumption becoming habitual rather than hunger-driven.
Cognitive Decline: DLPFC atrophy reduces the capacity for goal-directed eating, trapping individuals in automatic UPF consumption loops.
This interplay explains why UPF-driven obesity becomes self-perpetuating: metabolic dysfunction begets neurological changes reinforcing poor dietary choices. Breaking the cycle requires simultaneous targeting of hormonal signalling, neuroinflammation, and cognitive-behavioural pathways.
Impact of Obesity on metabolic, cognitive, immune and hormonal function, with associated disorders.
The Gut-Brain Axis: A Highway to Neuroinflammation
The gut-brain axis plays a critical role in the metabolic-neurological interface. Ultra-processed diets devastate gut ecology by reducing beneficial strains like Bifidobacterium and increasing pathogenic Proteobacteria.
The surge in LPS-producing pathogens fuels systemic inflammation, disrupting gut-brain signalling via the vagus nerve and inflaming the liver (disrupting the gut-liver-brain axis and affecting mood and cognition). The enteric nervous system suffers from SCFA depletion due to a lack of dietary (prebiotic) fibre, starving enteric neurones and leading to neuroinflammation.
Repairing the Metabolic-Neural Interface
Dietary countermeasures are essential to address metabolic dysfunction, systemic inflammation and neuroinflammation. Adopting a nutrient-dense diet rich in omega-3s and polyphenols can reduce BBB permeability and improve hippocampal neurogenesis. Time-restricted feeding (intermittent fasting), limiting eating to a 10-hour window, restores circadian lipid metabolism, lowering plasma FFAs. Pharmacological aids like GLP-1/GIP dual agonists promise to reduce UPF cravings by modulating mesolimbic dopamine pathways; however, they have been shown to have many long-lasting side effects.
Cognitive rehabilitation through working memory training and mindful eating protocols can also enhance dietary self-control by strengthening prefrontal inhibitory networks.
Metabolic and Cognitive Cross-Talk
The cycle of UPF consumption leading to adipose dysfunction, systemic inflammation, BBB breakdown, neuroinflammation, cognitive decline, and increased UPF intake is self-perpetuating. Breaking this cycle requires simultaneous targeting of gut microbiota, adipose plasticity, and neural repair mechanisms.
Conclusion:
The neurological fallout of UPF-driven obesity demands urgent paradigm shifts. Recognising adipose tissue as an endocrine organ and the BBB as a metabolic gatekeeper, we can develop interventions that restore lipid metabolism homeostasis, fortify neurovascular integrity, and rebalance gut-brain communication. Emerging research suggests that early intervention may prevent a substantial portion of UPF-related cognitive decline. Through combined strategies, we may mitigate this metabolic-neurological crisis’s rising tide.
Are you ready to become the hero of your own journey?
References
Avena, NM. Rada, P. Hoebel, BG. (2008). Evidence for sugar addiction: Behavioral and neurochemical effects of intermittent, excessive sugar intake. Neuroscience and Biobehavioral Reviews. 32(1), pp. 20-39. doi:10.1016/j.neubiorev.2007.04.019
Bleich, SN. Wolfson, JA. (2015). U.S. adults and child obesity trends, 1999-2014. American Journal of Preventive Medicine. 49(3), pp. 418-427. doi:10.1016/j.amepre.2015.05.006
Cordain, L. Eaton, SB. Sebastian, A. et al. (2005). Origins and evolution of the Western diet: Health implications for the 21st century. The American Journal of Clinical Nutrition. 81(2), pp. 341-354. doi:10.1093/ajcn/81.2.341
De Souza, RJ. Mente, A. Maroleanu, A. et al. (2015). Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: Systematic review and meta-analysis of observational studies. BMJ. 351, h3978. doi:10.1136/bmj.h3978
Flegal, KM. Kruszon-Moran, D. Carroll, MD. et al. (2016). Trends in obesity among adults in the United States, 2005 to 2014. JAMA. 315(21), pp. 2284-2291. doi:10.1001/jama.2016.6458
Hall, KD. Guo, J. (2017). Will calories be enough to tackle the obesity epidemic? American Journal of Clinical Nutrition. 106(3), pp. 751-755. doi:10.3945/ajcn.116.142328
Johnson, RK. Appel, LJ. Brands, MW. et al. (2009). Dietary sugars intake and cardiovascular health: A scientific statement from the American Heart Association. Circulation. 120(11), pp. 1011-1020. doi:10.1161/CIRCULATIONAHA.109.192627
Lustig, RH. (2010). Fructose: Metabolic, hedonic, and societal parallels with ethanol. Journal of the American Dietetic Association. 110(9), pp. 1307-1321. doi:10.1016/j.jada.2010.06.012
Monteiro, CA. Moubarac, JC. Cannon, G. et al. (2013). Ultra-processed products are becoming dominant in the global food system. Obesity Reviews. 14(2), pp. 21-28. doi:10.1111/obr.12065
Ng, SW. Slining, MM. Popkin, BM. (2014). Use of caloric and noncaloric sweeteners in US consumer packaged foods, 2005-2009. Journal of the Academy of Nutrition and Dietetics. 114(10), pp. 1548-1555.e6. doi:10.1016/j.jand.2014.05.014
Popkin, BM. Hawkes, C. (2016). Sweetening of the global diet, particularly beverages: Patterns, trends, and policy responses. The Lancet Diabetes & Endocrinology. 4(2), pp. 174-186. doi:10.1016/S2213-8587(15)00419-2
Schulze, MB. Hu, FB. (2005). Primary prevention of diabetes: What can be done and how much can be prevented? Annual Review of Public Health. 26, pp. 445-467. doi:10.1146/annurev.publhealth.26.021304.144532
Slavin, JL. Lloyd, B. (2012). Health benefits of fruits and vegetables. Advances in Nutrition. 3(4), pp. 506-516. doi:10.3945/an.112.002154
Swinburn, BA. Sacks, G. Hall, KD. et al. (2011). The global obesity pandemic: Shaped by global drivers and local environments. The Lancet. 378(9793), pp. 804-814. doi:10.1016/S0140-6736(10)62311-1
Taub, PR. Scherer, PE. (2013). Adiponectin: A key player in obesity related disorders. Current Pharmaceutical Design. 19(32), pp. 5765-5775. doi:10.2174/1381612811319320006
Wang, Y. Lobstein, T. (2006). Worldwide trends in childhood overweight and obesity. International Journal of Pediatric Obesity. 1(1), pp. 11-25. doi:10.1080/17477160600586747